Designed for Molecular Recycling: A Lignin-Derived Semi-aromatic Biobased Polymer
Özet
An example of a closed-loop approach towards a circular materials economy is described here. A whole sustainable cycle is presented where biobased semi-aromatic polyesters are designed by polymerization, depolymerization, and then re-polymerized with only slight changes in their yields or final properties.
Abstract
The development of chemically recyclable biopolymers offers opportunities within the pursuit of a circular economy. Chemically recyclable biopolymers make a positive effort to solve the issue of polymer materials in the disposal phase after the use phase. In this paper, the production of biobased semi-aromatic polyesters, which can be extracted entirely from biomass such as lignin, is described and visualized. The polymer poly-S described in this paper has thermal properties similar to certain commonly used plastics, such as PET. We developed a Green Knoevenagel reaction, which can efficiently produce monomers from aromatic aldehydes and malonic acid. This reaction has been proven to be scalable and has a remarkably low calculated E-factor. These polyesters with ligno-phytochemicals as a starting point show an efficient molecular recycling with minimal losses. The polyester poly(dihydrosinapinic acid) (poly-S) is presented as an example of these semi-aromatic polyesters, and the polymerization, depolymerization, and re-polymerization are described.
Introduction
In contrast to the incineration of polymeric waste, chemical recycling offers the possibility to recover the monomers. Chemical recycling is a logical choice at the end of the technical life of polymeric materials since these polymeric materials are produced chemically1. There are two ways to recycle the polymeric material chemically, pyrolysis and molecular recycling2. With pyrolysis, the polymeric material is transformed into products of higher value by using extreme conditions3,4. Molecular recycling is an efficient method for recovering the starting materials using depolymerization. After depolymerization, the monomeric units can be repolymerized into virgin polymeric materials5. The availability of suitable monomers to apply molecular recycling on a larger scale is wanting. The current plastic problem dictates that society demands sturdy and robust polymeric materials. At the same time, it is also preferred that the same polymeric materials are easily recyclable and do not endure in the environment. Current polymeric materials with good thermal and mechanical properties do not depolymerize easily6.
Lignin, commonly found in vascular plants, is responsible for 30% of the world's natural carbon content and is the second most abundant biopolymer after cellulose. Lignin has a complex amorphous structure and appears to be a suitable alternative to replace aromatics extracted from fossil materials. The three-dimensional structure of lignin provides strength and stiffness to wood, as well as resistance to degradation. Chemically speaking, lignin is a very complex polyphenolic thermoset. It consists of varying composition of three different methoxylated phenylpropane structures. Syringyl, guaiacyl, and p-hydroxyphenyl (often abbreviated as S, G, and H, respectively) are derived from the monolignols sinapyl alcohol, coniferyl alcohol, and p-coumaryl alcohol7. The distribution of these units differs per biomass type, with softwood, for instance, consisting of mostly guaiacyl units and hardwood of guaiacyl and syringyl units8,9. Renewable natural sources, such as trees and plants, are desirable for the production of redesigned monomers for innovative polymeric materials10. These monomers, isolated and synthesized from natural sources, are polymerized to so-called biobased polymers11.
Aromatic carboxylic acids are several orders of magnitude less electrophilic than the equivalent aliphatic carboxylic acids for electronic reasons12. Various commercial polyesters use aromatic carboxylic acids instead of aliphatic carboxylic acids. As a result, the fibers in polyester textiles made from poly(ethylene terephthalate) (PET) fibers are almost insensitive to hydrolysis during washing or, for example, rain13. When the molecular recycling of polyesters is wanted, it is advisable to use aliphatic esters in the build-up of the polymer.
For the reasons mentioned, we have investigated the possibilities of making polyesters from 4-hydroxy-3,5-dimethoxy-dihydrocinnamic acids14. Previous studies by Kricheldorf15, Meier16, and Miller17,18 show that it is challenging to build polymers using 4-hydroxy-3,5-dimethoxy-dihydrocinnamic acid. Decarboxylation and crosslinking hindered the polymerizations, and so limiting the success of these syntheses. Also, the mechanism of the polycondensation remained unclear. The presented paper describes the conditions in which the polyester poly(dihydrosinapinic acid) can be synthesized regularly and in high yield, thus paving the way for using semi-aromatic polyesters that are molecularly recyclable.
We have developed a green and efficient way to synthesize sinapinic acid using a condensation reaction between syringaldehyde and malonic acid19,20. After this Green Knoevenagel, hydrogenation produces dihydrosinapinic acid, which is suitable for reversible polycondensation. This publication visualizes the synthetic steps to the molecularly recyclable polymer poly(dihydrosinapinic acid), referring to the base units of lignin, called poly-S. After analyzing the polymeric material, poly-S is depolymerized to the monomer dihydrosinapinic acid under relatively favorable conditions and repolymerized over and over again.
Protocol
1. Green Knoevenagel condensation of syringaldehyde towards sinapinic acid with 5 mol% ammonium bicarbonate
- Add malonic acid (20.81 g, 200.0 mmol) together with syringaldehyde (36.4 g, 200.0 mmol) to a 250 mL round-bottom flask. Dissolve both constituents in 20.0 mL of ethyl acetate and add ammonium bicarbonate (790 mg, 10.0 mmol) to the flask.
NOTE: To ensure full completion of the condensation reaction, the rotary evaporator can be used to distill the ethyl acetate and concentrate the reaction mixture, resulting in a solvent-less reaction. - Keep the reaction mixture for 2 hours at 90 °C in the open flask without stirring for complete conversion to sinapinic acid.
NOTE: During this reaction, several morphological changes are observed. The reaction mixture changes from a dense grey mixture to a dissolved yellow mix due to the formation of water during the condensation step of the reaction. After evaporation of the condensate, the reaction mixture solidifies again, indicating full conversion. - In the work-up of the sinapinic acid product, dissolve the residue in 100 mL of a saturated aqueous NaHCO3-solution. Transfer the solution to a beaker and subsequently acidify it to a pH of 2 using 6 M HCl.
NOTE: While adding the saturated aqueous NaHCO3-solution, the crude product dissolves slowly and requires some time and manual scraping to dissolve fully. Once wholly dissolved and transferred to the beaker, the acidic solution of 6 M HCl is added drop-wise. Sinapinic acid will precipitate quickly, and the release of CO2 gas will cause the product to foam. - Separate the resulting residue by vacuum filtration and wash it with demineralized water.
NOTE: At this point, the purity of the product can be analyzed by HPLC. If the product appears contaminated, the next purification step (e.g., recrystallization) must be applied. - After recrystallization in a mixture of water-ethanol (4:1, v/v), separate the crystals by vacuum filtration. Dry the residue at 60 °C in a vacuum oven, resulting in 42.56 g of pure sinapinic acid.
NOTE: The purity of sinapinic acid is measured by melting point analysis and HPLC.
2. Hydrogenation of sinapinic acid towards dihydrosinapinic acid with RaneyTM nickel.
- Charge a 450 mL flask (using a hydrogenation apparatus) with sinapinic acid (33.6 g, 150 mmol). Dissolve the sinapinic acid in 300 mL of 2 M NaOH solution and add 1.5 g of nickel slurry (see Table of Materials) before fitting the flask to the apparatus.
NOTE: The color of the reaction mixture changes due to the addition of the alkaline solution. Sinapinic acid will be yellow in its crystalline form, but when deprotonated by the base, the color of the solution will change to red. The addition of nickel will turn the color black. - Pressurize the reactor with three bar of hydrogen gas and mechanically shake the reaction at 80 °C for three hours.
- Cool the reactor to room temperature and depressurized slowly. Recover most of the nickel catalyst with a magnet and subsequently filter the solution under reduced pressure.
NOTE: The reaction mixture should change from red to green color, visible after filtration. The notable color change is because the conjugated system is interrupted by hydrogenation of the double bond. - Acidify the solution with a 4 M HCl solution towards a pH of 2. Subsequently, perform an extraction with ethyl acetate (4 times 50 mL). Remove the solvent under reduced pressure after drying over MgSO4. Dry the solid product dihydrosinapinic acid at 60 °C in a vacuum oven.
NOTE: Theoretically, the polycondensation reaction can be processed on dihydrosinapinic acid, but there are some practical drawbacks. Due to the molecular structure, dihydrosinapinic acid sublimates above the melting point. An acetylation reaction can be performed to prevent sublimation and increase reactivity.
3. Acetylation of dihydrosinapinic acid towards acetylated monomers and oligomers (prepolymer).
- Add dihydrosinapinic acid (22.6 g, 100 mmol) to a 250 mL round-bottom flask, followed by acetic anhydride (14.2 mL, 150 mmol) and sodium acetate (0.82 g, 10 mmol).
NOTE: The dihydrosinapinic acid does not fully dissolve in the acetic anhydride at room temperature. - Heat the solution to 90 °C while stirring for one hour for complete conversion of dihydrosinapinic acid towards 4-acetoxy-dihydrosinapinic acid and its acetylated oligomers.
NOTE: All the products remain dissolved in acetic anhydride. To increase the total precipitation of 4-acetoxy-dihydrosinapinic acid, add a small amount of water-soluble solvent in the next step. - Dissolve the solid in 25 mL of acetone, precipitate (solvent-non solvent) into 250 mL of 0.1 M HCl while stirring vigorously and filter under vacuum.
NOTE: The best result shows an entirely precipitated white, sticky solid. If the product solution is added to the HCl solution too quickly, the product remains as a brown liquid at the bottom of the acidic solution. In this situation, extract the entire product solution with ethyl acetate (4 times 50 mL). After drying over MgSO4, remove the ethyl acetate under reduced pressure. The product is 4-acetoxy-dihydrosinapinic acid and prepolymer.
4. Polymerization of acetylated monomers and oligomers.
- Add the monomers, 4-acetoxy-dihydrosinapinic acid (20.8 g, 100 mmol), and prepolymer to a 100 mL round-bottom flask and add finely powdered NaOH (400 mg, 10.0 mmol). Heat the reaction mixture for three hours at a set temperature of 140 °C in the open flask while stirring at 100 rpm.
NOTE: During this reaction, water and acetic acid condense from the open flask. The pH of the vapor can be measured to confirm the condensation of acetic acid. The morphology of the reaction product changes from a molten state to a compact, light-brown product. The setup is subjected to a nitrogen flow to promote the evaporation of the condensate and prevent oxidation. Such reactions are usually stirred with a mechanical stirrer due to the high viscosity of the mixture. However, the use of a magnetical stirrer is sufficient, and the difference is negligible on this scale. - Set up a solvent assisted polymerization by adding zinc(II)acetate (180 mg, 1 mmol) and 25.0 mL of 1,2-xylene to the flask. Raise the set temperature to 160 °C.
NOTE: Raise the set temperature to 160 °C to ensure the reflux of 1,2-xylene and full reactivity of the polymerization. The temperature of the reaction itself remains at 144 °C, due to the boiling point of the 1,2-xylene. - Reflux the mixture at 144 °C for three hours with constant water and acetic acid removal using a Dean-Stark head.
NOTE: After adding the 1,2-xylene and heating the reaction flask to the required temperature, the reagents form a brown slurry with a lower viscosity. This lower viscosity is presumed to benefit the mobility of the end-groups of the polymer, promoting reactivity. - Cool the reaction mixture down and remove the 1,2-xylene by applying a vacuum (<10 mbar).
NOTE: The reaction mixture solidifies while evaporating the solvent and turns into an off-white solid. - Raise the temperature to 240 °C during the final stage of the polymerization and apply a high vacuum <1 mbar for thirty minutes.
NOTE: Only a slight amount of condensate evaporates during this step because only a small amount of condensation is needed to boost the chain length substantially at this stage of polymerization. - Cool the polymer poly-S to room temperature and wash with methanol to eliminate 4-acetoxy-dihydrosinapinic acid and prepolymers. The obtained product is a light-brown solid.
NOTE: After the process, the chain length and thermal properties of the polyester are investigated by GPC and DSC.
5. Representative procedure for the depolymerization of poly-S in 1 M NaOH:
- Finely grind and sieve poly-S into particles smaller than 180 μm to measure hydrolytic degradation.
NOTE: To grind the particles to the required size, use a mortar and pestle with liquid nitrogen-cooled poly-S, followed by a mechanical sieving step. - Load several test tubes with 20 mg of poly-S and add 1.0 mL of 1 M NaOH solution. Incubate the tubes at three different temperatures (RT, 50, and 80 °C), with an agitation of 500 rpm using a controlled environment incubator shaker.
NOTE: Use a heating-block/shaker with multiple entries for this reaction to ensure all the reactions take place in precisely the same (thermal) conditions. - Neutralize a test tube at regular time intervals with 1.0 mL of 0.5 M H2SO4 and add 2.0 mL of methanol after cooling.
NOTE: Small amounts of poly-S and only 1.0 mL of 1 M NaOH are used because these concentrations can, after the addition of methanol, be directly injected in HPLC and do not require any further dilution. - Filter all samples using 0.45 μm PTFE syringe filters and inject (20 μL) on HPLC using an autosampler. Monitor the absorbance at λ = 254 nm, and calculate the concentrations from a calibration curve of known dihydrosinapinic acid standard solutions.
NOTE: The standard solutions of the calibration curve should be made in the same solvent mixture as the work-up after the depolymerization.
Representative Results
Sinapinic acid was synthesized in high purity and high yield (> 95%) from syringaldehyde using the Green Knoevenagel condensation. (Supporting Information: Figure S1) The E-factor is an indication of waste production where a higher number indicates more waste. The E-factor is calculated by taking the total material input, subtracting the amount of the desired end product, and dividing the whole by the amount of the end product. This Green Knoevenagel condensation has an E-factor of 1.0, which can be calculated: [((29.81 g malonic acid + 36.4 g syringaldehyde + 0.790 g ammonium bicarbonate + 18.04 g ethyl acetate) – 42.56 g sinapinic acid) / 42.56 g sinapinic acid]. The sinapinic acid was hydrogenated with a high yield (>95%) to obtain the intended monomer dihydrosinapinic acid. (Supporting Information: Figure S2) This hydrogenation reaction has an E-factor of 0.84. Subsequently, dihydrosinapinic acid was acetylated to obtain 4-acetoxy-dihydrosinapinic acid and its prepolymer. The E-factor of this acetylation reaction was 0.45, assuming full conversion to 4-acetoxy-dihydrosinapinic acid (Supporting Information: Figure S3).
Usually, the E-factor of organic synthetic reactions of pharmaceuticals is between 5 and 100, and these reactions frequently use relatively harmful chemicals21. The E-factor, calculated for both the Green Knoevenagel and subsequent reactions, is remarkably low compared to these numbers. Yet, the E-factor exhibits some shortcomings. For example, the environmental hazard of a given waste component is not reflected by the E-factor. In principle, this can be compensated by the "environmental quotient" (Q) taking the "environmental unfriendliness" of a given waste into account. Because the catalyst ammonium bicarbonate is relatively harmless, the environmental unfriendliness quotient Q of the Green Knoevenagel is also stated as low. Still, additional research is essential to confirm this22. By applying the mentioned organic synthetic reactions, 4-acetoxy-dihydrosinapinic acid and its prepolymers are ready for the polycondensation to poly-S.
Figure 1 depicts the Proposed mechanism of the step-growth polymerization of 4-acetoxy-dihydrosinapinic acid. In step i, the added base starts the reaction and controls the reaction rate by deprotonating the acid group, thereby stimulating its reactivity23. The formed carboxylate attacks the carbonyl of the acetoxy group and creates a tetrahedral. In this state, the phenoxide group connects to the other carbonyl through a phenoxide shift, which was initially the aliphatic carboxylic acid group. When the phenoxide shift has occurred, the acetate-ion leaves the intermediate, subtracts a proton from the original base and then leaves the reaction as acetic acid. In practice, this step is promoted by creating a vacuum in the reaction vessel24. In each cycle, one novel molecule is added to the chain. During the progress of the polycondensation reaction, it turned out to be challenging to find the exact combination between increasing the molecular weight and degradation of the polymer chains. As the temperature was elevated with a gradient during the melt polymerization, growing amounts of degradation occurred as the viscosity of the polymer increased. A small amount of solvent was added during the second step of the polycondensation to decrease the viscosity. In this solvent-assisted poly-condensation, 1,2-xylene turns out to be a suitable agent for promoting the polycondensation25.
Figure 2 represents three different 1H NMR measurements during the polymerization process. The samples dissolved well and were measured in a mixture of CF3COOD / CDCl3. The 1H NMR spectra were taken after the acetylation of sinapinic acid. The 1H NMR spectrum in the middle is a sample taken from the same reaction. No specific compound was isolated, so acetylated sinapic acid and its prepolymers are shown. The 1H NMR spectrum above was taken from a sample after completion of the polycondensation. The areas of the acetyl end groups decrease during the polymerization process, and the number average degree of polymerization (DPn) increases. After completing the reaction, the degree of polymerization was set at 43 repeating units as determined by 1H NMR measurements.
GPC measurements were performed to investigate the chain length of poly-S, and various GPC-systems have been tested for the analysis. The initial step was to dissolve the polymeric material in several common organic solvents such as chloroform (CHCl3), dimethylformamide (DMF), hexafluoro-2-propanol (HFIP), and tetrahydrofuran (THF). However, it appears that the highly crystalline material of poly-S, visible on DSC thermograms, was sparingly soluble in these solvents. Also, when the polymeric material was melted and then cooled very quickly in liquid nitrogen to increase the amorphicity of the polymer, only the short polymer chains dissolved in DMF. The value measured on GPC was only 1,900 up to 2,100 Da, which corresponds to approximately ten repeating units.
Another pretreatment method has been selected to perform a proper GPC measurement. Additional research26 prescribes that a more amorphous polyester material can be formed when polystyrene in its atactic form is melt-mixed into the polyester. The two polymers (polyester and polystyrene) were placed together in a DSC pan, melted, and subsequently cooled in liquid nitrogen. Washing the quenched mixture with acetone removed the residual polystyrene, and after successfully dissolving in DMF, a high molecular weight polymer could be observed.
When poly-S was hydrolyzed in 1 M NaOH, the molecularly recyclable polyester yielded the starting material dihydrosinapinic acid in less than 10 minutes. HPLC, melting point analysis, and 1H NMR confirmed this observation (Supporting information: Figure S4). Extended reaction times did not increase the yield, as shown in Figure 3.
The thermograms and GPC analysis of poly-S 1.0 and their successive generations poly-S 2.0 and poly-S 3.0 after forced amorphicity with atactic polystyrene are shown in Figure 4 and Table 1, respectively. DSC analysis of poly-S shows a glass transition signal (Tg) at 113 °C and an endothermic melting signal (Tm) at 281 °C. The polymers from re-polymerized sinapinic acid 3a poly-S 2.0 up and poly-H 3.0 exhibit similar thermal properties (Supporting Information: Figures S5-S7). Poly-S is a semi-crystalline polymeric material because glass transition temperatures (Tg) and melting signals are present in the thermograms of poly-S and their successive generations. It can be stated GPC analysis after forced amorphicity with atactic polystyrene displays a constant length of polymeric material throughout the different generations (Supporting Information: Figures S8-S10).
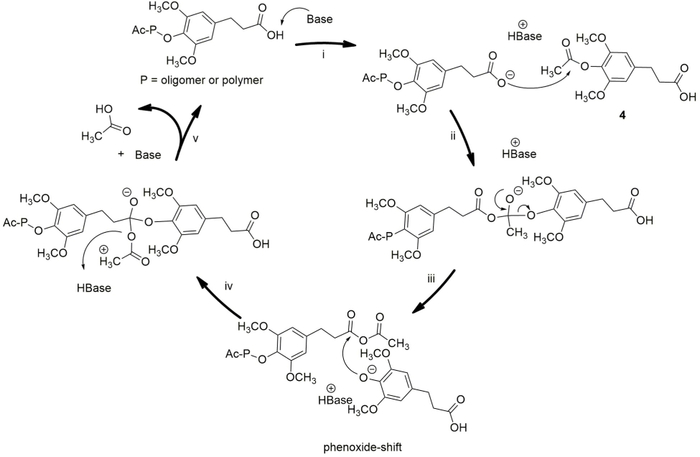
Figure 1: Proposed mechanism of the step-growth polycondensation of 4-acetoxy-dihydrosinapinic acid. Please click here to view a larger version of this figure.
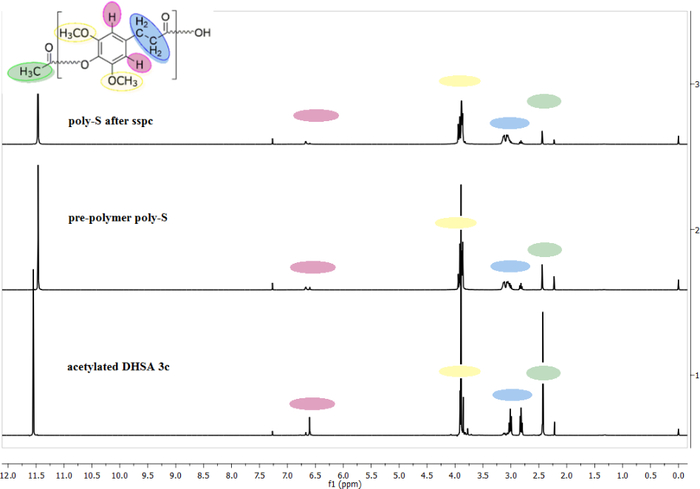
Figure 2: Overlay of 1H NMR spectra (25 °C, CF3COOD/CDCl3, with residual solvent peaks at 11.49 and 7.27 ppm for CF3COOH and CHCl3, respectively) of the isolated acetylation product of sinapinic acid (bottom); the prepolymer of poly-S (middle); poly-S after solid-state post condensation (240 °C, 30 min, vacuum) (top). Please click here to view a larger version of this figure.
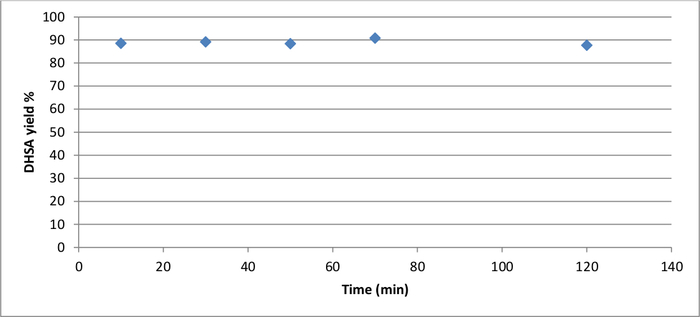
Figure 3: Alkaline hydrolysis (1 M NaOH solution, 80 °C) of poly-S over time, determined by dihydrosinapinic acid (DHSA) yield based on HPLC peak areas. Please click here to view a larger version of this figure.

Figure 4: DSC thermogram of three generations poly-S. Please click here to view a larger version of this figure.
| Generation | Poly-S (mg) | PS (mg) | Mn | Mw | PDI | DP |
| 1.0 | 5.0 | 4.8 | 120030 | 193580 | 1.6 | 594 |
| 2.0 | 5.0 | 5.0 | 134740 | 194410 | 1.4 | 667 |
| 3.0 | 4.7 | 4.9 | 153620 | 237210 | 1.5 | 760 |
Table 1: GPC measurements of poly-S generations.
Discussion
When dihydrosinapinic acid was heated in a reaction vessel, sublimation of the starting material occurred, and this effect was enhanced when a vacuum was applied. Acetylation has been performed on dihydrosinapinic acid to avoid sublimation. Kricheldorf et al.12,27 recognized that not only acetylation but similarly di- and oligomerization occurred. However, these esterified monomers and oligomers no longer sublimate and are suitable as monomers for the melt polycondensation28. Also, the formation of the prepolymers from 4-acetoxy-dihydrosinapinic acid confirms the hypothetical reaction mechanism29.
1H NMR measurements were used to determine the number average degree of polymerization, and GPC measurements were used to generate further chain length data. GPC measurements were always performed after forcing amorphicity by mixing polystyrene and quenching with liquid nitrogen.
The obtained results presented in Table 1 show the GPC Mn values of the polymer samples correspond to about 670 repeating units of poly-S, on average, throughout the generations. These values are considerably higher than the corresponding values obtained with 1H NMR measurements presented in Figure 2 since these values are about 43 repeating units of poly-S. This variation is related to the trace impurities of the acetoxy-group containing compounds within the synthesized polymers, decreasing the number average degree of polymerization (DPn) of poly-S, calculated with 1H NMR measurements. Another explanation is related to the differences in hydrodynamic volumes of the polyesters and polystyrene standards used during the GPC measurements. The polydispersity PDI is below 2.0. This relatively low value can be attributed to high molecular polymer filtering out before GPC analysis.
Thermal characterization was carried out by using DSC and TGA techniques, whereby thermal properties were found comparable to PET. DSC analysis showed that the molecularly recyclable polymer poly-S has a melting temperature of 281 °C. This melting temperature is sixteen degrees higher related to PET30. The glass-rubber transition temperatures of poly-S are also higher compared to PET, namely 113 °C versus 67 °C with PET. A higher glass-rubber transition temperature is often beneficial for many applications.
Finally, during the cooling cycle, poly-S has a lower crystallization temperature related to PET, namely 151 °C versus 190 °C. The thermal gravimetrical analysis of poly-S indicated a decomposition temperature of 341 °C of poly-S. Compared to PET with a decomposition temperature of 470 °C, the decomposition temperature of poly-S is lower31 (Supporting Information: Figure S11).
The depolymerization of the formed polymeric material was observed for over two hours, at a temperature of 80 °C. HPLC-samples were taken to analyze the concentration of released dihydrosinapinic acid in a solution of 1 M NaOH. The samples were measured against a calibration curve of pure dihydrosinapinic acid to determine the HPLC yield. (Supporting Information: Figure S12)
A mass balance has been established to determine the loss of mass during a complete cycle of polymerization and depolymerization. An exact amount of 5.00 grams of dihydrosinapinic acid has been introduced into the polymerization process. After one cycle of polymerization, depolymerization to dihydrosinapinic acid, and work-up, 4.70 grams of dihydrosinapinic acid was produced, which corresponds to a yield of 94%. During the first cycle, most of the material was lost by manual handling (Supporting Information: Figure S13-S14). The loss of the monomer dihydrosinapinic acid was reasonably limited so that after three generations, approximately 85% of the initial amount of monomer remained.
This publication describes how a new polymeric material that is specifically suitable for molecular recycling has been designed. The combination of polymerization, depolymerization, in which the monomers are recovered, and repolymerization, has created an utterly sustainable cycle with only minimal losses. When the principles of Green Chemistry are applied further, it is possible to optimize the conditions of different steps. For example, subsequent studies can investigate hydrolysis under acidic conditions to prevent excessive salt-formation.
Açıklamalar
The authors have nothing to disclose.
Acknowledgements
The authors are grateful for the financial support from the Netherlands Organization for Scientific Research (NWO) (grant 023.007.020 awarded to Jack van Schijndel).
Materials
| Reaction 1: Green Knoevenagel condensation | |||
| Ammonium bicarbonate | Sigma Aldrich | >99% | |
| Ethanol | Boom | Technical grade | |
| Ethyl acetate | Macron | 99.8% | |
| Hydrochloric acid | Boom | 37% | |
| Malonic acid | Sigma Aldrich | 99% | used as received |
| Sodium bicarbonate | Sigma Aldrich | >99.7% | |
| Syringaldehyde | Sigma Aldrich | 98% | used as received |
| Reaction 2: Hydrogenation | |||
| Magnesium sulfate | Macron | 99% | dried |
| Raney™ nickel | Sigma Aldrich | >89% | |
| Sodium hydroxide | Boom | Technical grade | dissolved |
| Reaction 3: Acetylation | |||
| Acetic anhydride | Macron | >98% | |
| Acetone | Macron | >99.5% | |
| Sodium acetate | Sigma Aldrich | >99% | |
| Reaction 4A: Polymerisation | |||
| 1,2-xylene | Macron | >98% | |
| Sodium hydroxide | Boom | Technical grade | finely powdered |
| Zinc(II)acetate | Sigma Aldrich | 99.99% | |
| Reaction 4B: Depolymerisation | |||
| Sodium hydroxide | Boom | Technical grade | dissolved |
| Sulfuric acid | Macron | 100% | |
| Analysis | |||
| CDCl3 | Cambride Isotope Laboratories, Inc. | 99.5% | |
| CF3COOD | Cambride Isotope Laboratories, Inc. | 98% | |
| Dimethylformamide | Macron | >99.9% | |
| Hexafluoro-2-propanol | TCI Chemicals | >99% | |
| Methanol | Macron | >99.8% | |
| Tetrahydrofuran | Macron | >99.9% |
Referanslar
- Rahimi, A., García, J. M. Chemical recycling of waste plastics for new materials production. Nature Reviews Chemistry. 1 (6), 41570 (2017).
- Sardon, H., Dove, A. Plastics recycling with a difference. Science. 360 (6387), 380-381 (2018).
- Jones, G. O., Yuen, A., Wojtecki, R. J., Hedrick, J. L., García, J. M. Computational and experimental investigations of one-step conversion of poly(carbonate)s into value-added poly(aryl ether sulfone)s. Proceedings of the National Academy of Sciences of the United States of America. 113 (28), 7722-7726 (2016).
- García, J. M., et al. Recyclable, Strong Thermosets and Organogels via Paraformaldehyde Condensation with Diamines. Science. 344 (6185), 1251484 (2014).
- Brutman, J. P., De Hoe, G. X., Schneiderman, D. K., Le, T. N., Hillmyer, M. A. Renewable, Degradable, Chemically Recyclable Cross-Linked Elastomers. Industrial & Engineering Chemistry Research. 55 (42), 11097-11106 (2016).
- Hong, M., Chen, E. Y. Completely recyclable biopolymers with linear and cyclic topologies via ring-opening polymerization of γ-butyrolactone. Nature Chemistry. 8 (1), 42-49 (2016).
- Lochab, B., Shukla, S., Varma, I. K. Naturally occurring phenolic sources: monomers and polymers. RSC Advances. 4 (42), 21712 (2014).
- Fache, M., Boutevin, B., Caillol, S. Vanillin, a key-intermediate of biobased polymers. European Polymer Journal. 68, 488-502 (2015).
- Pinto, P. C. R., Costa, C. E., Rodrigues, A. E. Oxidation of Lignin from Eucalyptus globulus Pulping Liquors to Produce Syringaldehyde and Vanillin. Industrial & Engineering Chemistry Research. 52 (12), 4421-4428 (2013).
- Llevot, A., Grau, E., Carlotti, S., Grelier, S., Cramail, H. From Lignin-derived Aromatic Compounds to Novel Biobased Polymers. Macromolecular Rapid Communications. 37 (1), (2016).
- Hernández, N., Williams, R. C., Cochran, E. W. The battle for the “green” polymer. Different approaches for biopolymer synthesis: bioadvantaged vs. bioreplacement. Organic & Biomolecular Chemistry. 12 (18), 2834-2849 (2014).
- Kricheldorf, H. R., Stukenbrock, T. New polymer syntheses 85. Telechelic, star-shaped and hyperbranched polyesters of β-(4-hydroxyphenyl) propionic acid. Polymer. 38 (13), 3373-3383 (1997).
- Gilding, D. K., Reed, A. M. Biodegradable polymers for use in surgery-poly(ethylene oxide) poly(ethylene terephthalate) (PEO/PET) copolymers: 1. Polymer. 20 (12), 1454-1458 (1979).
- Jiang, Y., Loos, K. Enzymatic Synthesis of Biobased Polyesters and Polyamides. Polymers. 8 (7), 243 (2016).
- Kricheldorf, H. R., Stukenbrock, T. New polymer syntheses, 92. Biodegradable, thermotropic copolyesters derived from β-(4-hydroxyphenyl)propionic acid. Macromolecular Chemistry and Physics. 198 (11), 3753-3767 (1997).
- Kreye, O., Oelmann, S., Meier, M. A. Renewable Aromatic-Aliphatic Copolyesters Derived from Rapeseed. Macromolecular Chemistry and Physics. 214 (13), 1452-1464 (2013).
- Mialon, L., Pemba, A. G., Miller, S. A. Biorenewable polyethylene terephthalate mimics derived from lignin and acetic acid. Green Chemistry. 12 (10), 1704 (2010).
- Nguyen, H. T. H., Reis, M. H., Qi, P., Miller, S. A. Polyethylene ferulate (PEF) and congeners: polystyrene mimics derived from biorenewable aromatics. Green Chemistry. 17 (9), 4512-4517 (2015).
- van Schijndel, J., Canalle, L. A., Molendijk, D., Meuldijk, J. The Green Knoevenagel Condensation: Solvent-free Condensation of Benzaldehydes. Green Chemistry Letters and Reviews. 10 (4), 404-411 (2017).
- van Schijndel, J., Molendijk, D., Spakman, H., Knaven, E., Canalle, L. A., Meuldijk, J. Mechanistic considerations and characterization of ammonia-based catalytic active intermediates of the Green Knoevenagel reaction of various benzaldehydes. Green Chemistry Letters and Reviews. 12 (3), 323-331 (2019).
- Sheldon, R. A. Fundamentals of green chemistry: efficiency in reaction design. Chemical Society Reviews. 41 (4), 1437-1451 (2012).
- van Schijndel, J., Molendijk, D., Canalle, L. A., Rump, E. T., Meuldijk, J. Temperature Dependent Green Synthesis of 3-Carboxycoumarins and 3,4-unsubstituted Coumarins. Current Organic Synthesis. 16 (1), 130-135 (2019).
- Bloom, M. E., Vicentin, J., Honeycutt, D. S., Marsico, J. M., Geraci, T. S., Miri, M. J. Highly renewable, thermoplastic tetrapolyesters based on hydroquinone, p-hydroxybenzoic acid or its derivatives, phloretic acid, and dodecanedioic acid. Journal of Polymer Science Part A: Polymer Chemistry. 56 (14), 1498-1507 (2018).
- Padias, A. B., Hall, H. K. Mechanism Studies of LCP Synthesis. Polymers. 3 (2), 833-845 (2011).
- Moon, S., Lee, C., Taniguchi, I., Miyamoto, M., Kimura, Y. Melt/solid polycondensation of l-lactic acid: an alternative route to poly(l-lactic acid) with high molecular weight. Polymer. 42 (11), 5059-5062 (2001).
- Wellen, R. M. R. Effect of polystyrene on poly(ethylene terephthalate) crystallization. Materials Research. 17 (6), 1620-1627 (2014).
- Kricheldorf, H. R., Conradi, A. New polymer syntheses 16. LC-copolyesters of 3-(4-hydroxyphenyl) propionic acid and 4-hydroxy benzoic acids. Journal of Polymer Science Part A: Polymer Chemistry. 25 (2), 489-504 (1987).
- Chen, Y., Tan, L., Chen, L., Yang, Y., Wang, X. Study on biodegradable aromatic/aliphatic copolyesters. Brazilian Journal of Chemical Engineering. 25 (2), 321-335 (2008).
- Han, X., et al. A Change in Mechanism from Acidolysis to Phenolysis in the Bulk Copolymerization of 4-Acetoxybenzoic Acid and 6-Acetoxy-2-naphthoic Acid. Macromolecules. 29 (26), 8313-8320 (1996).
- Lu, X. F., Hay, J. N. Isothermal crystallization kinetics and melting behaviour of poly(ethylene terephthalate). Polymer. 42 (23), 9423-9431 (2001).
- Sánchez, M. E., Morán, A., Escapa, A., Calvo, L. F., Martínez, O. Simultaneous thermogravimetric and mass spectrometric analysis of the pyrolysis of municipal solid wastes and polyethylene terephthalate. Journal of Thermal Analysis and Calorimetry. 90 (1), 209-215 (2007).

