Synthesizing Lipid Nanoparticles by Turbulent Flow in Confined Impinging Jet Mixers
Summary
A detailed protocol for synthesizing lipid nanoparticles (LNPs) using confined impinging jet (CIJ) mixer technologies, including a two-jet CIJ and a four-jet multi-inlet vortex mixer (µMIVM), is demonstrated. The CIJ mixers generate reproducible, turbulent micro-mixing environments, resulting in the production of monodisperse LNPs.
Abstract
Lipid nanoparticles (LNPs) have demonstrated their enormous potential as therapeutic delivery vehicles, as evidenced by the approval and global usage of two COVID-19 messenger RNA (mRNA) vaccines. On a small scale, LNPs are often made using microfluidics; however, the limitations of these devices preclude their use on a large scale. The COVID-19 vaccines are manufactured in large quantities using confined impinging jet (CIJ) turbulent mixers. CIJ technology enables production at a laboratory scale with the confidence that it can be scaled to production volumes. The key concepts in CIJ mixing are that the mixing length and time scale are determined by the turbulence intensity in the mixing cavity and that the nanoparticle formation occurs away from walls, eliminating the problem of deposition on surfaces and fouling. This work demonstrates the process of making LNPs using confined impinging jet mixer technology with two geometries: the two-jet CIJ and the four-jet multi-inlet vortex mixer (MIVM). The advantages and disadvantages of each mixing geometry are discussed. In these geometries, LNPs are formed by rapid mixing of an organic solvent stream (usually ethanol containing the ionizable lipids, co-lipids, and stabilizing PEG-lipids) with an aqueous anti-solvent stream (aqueous buffer containing RNA or DNA). The operating parameters for the CIJ and MIVM mixers are presented to prepare reproducible LNPs with controlled size, zeta potential, stability, and transfection effectiveness. The differences between LNPs made with poor mixing (pipetting solutions) compared to CIJ mixing are also presented.
Introduction
mRNA-based therapeutics hold great potential for the treatment and prevention of a wide range of diseases, including infectious diseases, genetic disorders, and cancers1. Unlike small-molecule therapeutics, which can passively diffuse across the cell membrane, nucleic acids must be encapsulated for intracellular delivery2. Encapsulation provides both structure and stability to mRNA, facilitating their intracellular delivery via endocytic pathways as well as preventing degradation from intra- and extra-cellular components such as nucleases3. A host of materials and nanocarriers have been developed for the encapsulation and delivery of mRNA, including inorganic nanoparticles, polymers, lipids, and lipid-like materials1. Among these, LNPs have emerged as the most prominent delivery platform for mRNA-based therapeutics4.
LNPs are composed of four lipid components: ionizable lipid, cholesterol, zwitterionic lipid, and PEG-lipid stabilizer5. Ionizable lipids suitable for mRNA delivery exhibit a careful balance between the lipid hydrophobicity and the dissociation constant (pKa) of a ternary amine group6. The ionizable lipid pKa typically has a pH between 6.0 and 6.7, such as KC-2 (DLin-KC2-DMA), MC-3 (DLin-MC3-DMA), and ALC-03157. This pKa restriction on the ionizable lipid enables both the encapsulation of nucleic acid polymers as hydrophobic lipid salts and the intracellular delivery through an "endosome escape" process. LNPs enter a target cell through (various) endocytosis pathways that all involve acidification of the endosome from pH 7.4 to pH ~58. The ionizable lipid pKa ensures that LNPs have nearly neutral surfaces under physiological conditions but become cationic in an acidifying endosome9. This pH response enables selective disruption of only the endosomal membrane, the release of the encapsulated nucleic acid polymer, and preserves cell viability, unlike permanently cationic lipids used in transfection systems such as Lipofectamine. Cholesterol is a hydrophobic, interstitial molecule in the LNP structure that improves lipid fluidity. The zwitterionic lipid plays a structural role and forms a bilayer on the LNP surface. The poly(ethylene glycol)-lipid (PEG-lipid) is a colloidal stabilizer that enhances the LNP stability by imparting a polymeric steric stabilizer onto the LNP surface, which resists aggregation of LNPs. This stabilizes the LNP, particularly during changes in pH that regenerate the free base form of the ionizable lipid which behaves like a hydrophobic oil. The Onpattro (patisiran) recipe (hereafter, referred to as LNP formulation) is often used as a starting point for LNP formulation with ionizable lipid MC3, cholesterol, distearoylphosphatidylcholine (DSPC), and PEG2000-DMG dissolved in ethanol mixed against an aqueous solution of RNA10.
Several techniques can be used to manufacture LNPs encapsulating nucleic acid polymers, with most of them relying on a common theme of rapidly mixing an ethanol stream containing lipids with an aqueous stream incorporating the nucleic acid of interest (siRNA, mRNA, or DNA)9,11,12,13,14. In this regard, bulk mixing processes such as pipette mixing and vortex mixing offer a simple strategy to form LNPs that eliminates the need for using sophisticated instruments12. However, bulk mixing does not provide a homogeneous distribution of components, leading to a sub-optimal LNP size distribution along with significant batch-to-batch variability15.
Laboratories routinely use microfluidic mixing techniques to obtain reproducible LNPs by achieving more precise control over mixing conditions12,13,16. Yet, the laminar flow conditions in microfluidic devices, which are inherent due to the small length scales and low velocities in a microfluidics chamber, result in comparatively slow solvent/antisolvent mixing17. The small chamber dimensions severely limit the throughput and scalability needed for GMP production of LNPs, but researchers have parallelized microfluidic chambers to attempt to scale production volumes15. A parallelized microfluidics geometry does not eliminate the problem of lipid adsorption to surfaces during large volume processing, a problem commonly referred to as "fouling" of the mixing device, and there are problems with uniformity and stability of flows that make microfluidics scaleup challenging for industrial-scale production18,19. It is not surprising that pharmaceutical companies used turbulent impinging jet mixers to manufacture COVID-19 vaccination mRNA-LNPs20.
The production process of RNA-loaded LNPs entails the blending of an aqueous buffer stream containing the RNA payload with an ethanol stream containing the four distinct lipid components. These formulations utilize an acidic buffer with a pH of 4.0 or less, which charges the ionizable lipid as the aqueous and ethanolic streams mix. The positively charged ionizable lipids interact electrostatically with the negatively charged RNAs, forming a hydrophobic RNA-lipid salt. Hydrophobic lipid species, including the RNA-lipid salt, precipitate in the mixed solvents and form hydrophobic nuclei. These nuclei grow through the precipitation of zwitterionic lipid and cholesterol until reaching a critical point where sufficient pegylated lipid adsorbs on the surface of the LNPs, halting further growth -nucleation and growth mechanism21,22,23. The addition of aqueous buffer to the lipid solution, to the extent where lipids precipitate and LNPs form, hinges on two distinct time scales: the solvent-antisolvent mixing period, τmix, and the nuclei growth period, τagg. The dimensionless Damköhler number, defined as Da = τmix/τagg, captures the interplay between these time scales24. In instances of slow mixing (Da > 1), the final size of LNPs is transport-controlled and varies with mixing time. Conversely, during fast mixing (Da < 1), the fluid is fragmented into Kolmogrov-length striations or layers, whereby LNP formation is solely governed by the molecular diffusion of each constituent, resulting in homogeneous kinetics of LNP formation. Achieving the latter scenario demands that the lipid concentration exceeds a critical threshold, establishing a state of supersaturation conducive to uniform homogeneous nucleation.
It is estimated that τagg ranges from a few tens to a few hundreds of milliseconds25. In its most basic configuration, the two streams, one containing ethanol with lipids and the other containing an aqueous buffer with RNA cargo, are injected into a chamber known as a "confined impinging jet" (CIJ) mixer. Turbulent vortices produce solvent/antisolvent striation length scales of 1 µm within 1.5 ms when operated at appropriate velocities. The stream velocities and mixing geometry determine the conversion of linear momentum into turbulent vortices that mix the streams. This is parameterized by the dimensionless number, the Reynolds number (Re), that is linearly proportional to the flow velocities. Re is computed from Re = Σ (ViDi/vi), where Vi is the flow velocity in each steam, vi is the kinematic viscosity of each stream, and Di is the stream inlet diameter in 2-jet CIJ devices26 or the chamber diameter in 4-jet MIVMs27. Note: Some references for the CIJ use only a single jet diameter and velocity to define Re28. Re is in the range of 1-100 in a microfluidics device, whereas in the CIJ devices, Re of 125,000 can be achieved. In a CIJ mixer, streams with equal momentum collide, dissipating their momentum upon impact as turbulent mixing, which leads to efficient micromixing due to the small Kolmogorov microscales and small Damköhler number. Another type of mixer is the "multiple inlet vortex mixer" (MIVM), where four streams are directed into a central chamber. In this setup, continuous flows into the confined mixing chamber ensure a well-defined mixing time scale. All fluid elements pass through the high-energy mixing zone in both types of mixers. In contrast, simple mixing devices like T-junctions do not contain a chamber that provides a mixing zone, resulting in less mixing of the two streams due to the incoming stream momentum being largely deflected into the outlet direction rather than into turbulent vortex generation. Both CIJ and MIVM mixers can be operated in batch or continuous modes, offering flexibility for LNP production at various scales.
This protocol describes how optimal LNP formulations are made by employing two confined impinging jet technologies: 2-jet CIJ and the 4-jet MIVM mixers. The operation of CIJ and MIVM mixers has been previously demonstrated for the preparation of NPs with hydrophobic core materials29. That article and video should be consulted as an additional resource on the formation of NPs with these mixers. This update focuses on lipid-based NP formation. The ability to tune the size of LNPs by varying the micro-mixing conditions is demonstrated. Additionally, the utility of CIJ technologies in forming stable, monodisperse LNPs with improved in vitro transfection efficiencies in HeLa cells when compared to LNPs made using poor pipette mixing is shown. Furthermore, the advantages and disadvantages of each CIJ mixing geometry, along with appropriate conditions needed for the scaleup of these mixers, are discussed.
Protocol
The details of the reagents and the equipment used in this study are listed in the Table of Materials.
1. Preparation of buffers, solvent and antisolvent streams
- Dissolve all lipid components in ethanol at given mass concentrations, as shown in Table 1, to make a patisiran LNP formulation10. Prior to use, heat the solutions to 37 °C with gentle sonication to re-dissolve any precipitated solids.
NOTE: Larger stock solutions of several milliliters can be prepared and stored at 4 °C. - Prepare an acetate buffer stock solution at a concentration of 100 mM, pH 4, by combining 186 mg of sodium acetate and 464 mg of acetic acid in 80 mL of nuclease-free water. Adjust the pH with concentrated HCl or NaOH as needed and make up the final volume to 100 mL.
- Prepare a HEPES buffer stock solution at a concentration of 1 M, pH 7.5, by dissolving 23.82 g of HEPES salt in 80 mL of nuclease-free water. Adjust the pH with concentrated HCl or NaOH as needed and make up the final volume to 100 mL.
- Dilute the nucleic acid polymer of interest and the 100 mM acetate buffer stock with nuclease-free water to produce a 300 µg/mL solution of RNA in 30 mM acetate buffer. Prepare an adequate volume of working solution for the designed experiments, and this protocol requires 1500 µL total for both the CIJ and MIVM mixers.
NOTE: RNA obtained from the supplier will already be in an appropriate buffer, typically at a concentration of 10 mg/mL (yeast RNA) or 1 mg/mL (luciferase-encoding mRNA, LucRNA, or GFP-encoding mRNA, GFP-RNA). Yeast RNA may be used as a low-cost model RNA for precipitations.
2. Formulation of LNPs using a two-jet CIJ mixer
- Preparation and cleaning of equipment
- Clean CIJ mixers immediately before use by flushing the mixer with ethanol. Fill two 5 mL syringes with ethanol and lock each into an inlet port of the CIJ. Rapidly depress the syringes and collect the mixer effluent as waste.
NOTE: CIJ mixers may be constructed and operated as previously described29. Various support stands may be used to hold the CIJ mixer, including a ring stand, centrifuge tube holder, or Erlenmeyer flask. The CIJ suppliers are listed in the Table of Materials and the Supplementary File 1. - Remove the ethanol flush syringes from the CIJ. These syringes may be stored and reused for additional ethanol rinses of the CIJ.
- Dry the internal channels of CIJ by blowing a dry nitrogen stream through the inlet adapters. Use only dry, filtered air that is not contaminated with pump oil mist or aerosols if nitrogen is not available.
- Clean CIJ mixers immediately before use by flushing the mixer with ethanol. Fill two 5 mL syringes with ethanol and lock each into an inlet port of the CIJ. Rapidly depress the syringes and collect the mixer effluent as waste.
- Preparation of solvent and antisolvent streams
- Mix the lipid stock solutions and dilute with additional ethanol to produce 500 µL of the 6 mg/mL lipid solution listed in Table 1 in a 1.5 mL microcentrifuge tube.
NOTE: This formulation is the commonly used Onpattro composition containing 50 mole% MC3, 10 mole% DSPC, 38.5 mole% cholesterol, and 1.5 mole% DMG-PEG2000. - Dilute the 100 mM pH 4 acetate buffer stock to 10 mM in a total volume of 4 mL as the CIJ mixer effluent quench bath.
NOTE: A magnetic stir bar may also be added to the quench bath. - Withdraw 500 µL of RNA solution prepared in step 1.4 in a 1 mL syringe. Invert the syringe and expel any air from this aqueous antisolvent stream containing nucleic acid polymer.
- Withdraw 500 µL of the lipid solution prepared in step 2.2.1 in a 1 mL syringe. Invert the syringe and expel any air from this ethanolic solvent stream containing lipids.
- Mix the lipid stock solutions and dilute with additional ethanol to produce 500 µL of the 6 mg/mL lipid solution listed in Table 1 in a 1.5 mL microcentrifuge tube.
- LNP production in a CIJ mixer
- Position the clean CIJ mixer over the quench bath vial.
NOTE: A ring stand clamp or tube rack makes a convenient support for the CIJ mixer. See Figure 1A for a typical CIJ setup. - Mate the two syringes filled in step 2.2.3 and step 2.2.4 to the CIJ mixer inlet ports.
- Depress both syringes rapidly to mix the solvent and antisolvent streams and collect lipid NPs in the quench bath.
NOTE: the syringes must be advanced quickly (in under 1 s) but smoothly and uniformly. Asymmetric flow or slow flow will produce polydisperse, large LNPs. - Remove the CIJ mixer from over the quench bath with the syringes still attached.
NOTE: Do not remove the syringes or allow the hold-up volume to flow into the quench bath, as this material is poorly mixed and will adversely impact the production dispersion properties if mixed into the quench bath. - Hold the CIJ over a waste container and remove the syringes, allowing the residual hold-up volume to flow into the waste container. Dispose these syringes and repeat the cleaning process as described in section 2.1.
- Analyze LNPs produced with the CIJ as described in section 5 below.
- Position the clean CIJ mixer over the quench bath vial.
3. Formulation of LNPs using a four-jet MIVM mixer
- Assembly of a MIVM mixer, referred to as a micro-size MIVM (µMIVM) to distinguish it from larger models used for scaled-up production
- Gather all the individual components required to assemble a MIVM mixer: the bottom receiver, the mixing geometry disk, the top disk, the O-ring, and the spanner wrench.
NOTE: MIVM construction, assembly, and operation have been previously demonstrated for the encapsulation of hydrophobic species, and the suppliers are listed in Supplementary File 1 and the Table of Materials29. See Figure 2 for a schematic of components and mixer stand terminology. - Seat the O-ring into the groove in the mixing disk.
- Align the mixing disk holes with the pegs on the top disk and push them together without dislodging the O-ring.
- Screw the mated mixing disk, O-ring, and top disk assembly into the bottom receiver loosely.
NOTE: Remove the outlet tubing from the bottom receiver before this step. - Tighten the top disk into the bottom receiver using the spanner wrench.
NOTE: A food or pharmaceutical-grade anti-seize compound may be applied to the top disk threads if thread galling is observed. - Insert and tighten the outlet tubing fitting into the bottom receiver to complete the MIVM.
- Mount the assembled MIVM into the mixer stand so the outlet tubing exits through the support plate.
NOTE: The procedure for adjusting the MIVM mixer stand should be periodically performed to make sure the mechanical stops on the mixer stand are correctly configured29.
- Gather all the individual components required to assemble a MIVM mixer: the bottom receiver, the mixing geometry disk, the top disk, the O-ring, and the spanner wrench.
- Preparation of solvent and antisolvent streams
- Mix the two ethanolic lipid solvent stream solutions in a 1.5 mL microcentrifuge tube, as given in Table 2, by diluting the lipid stock solutions and adding additional ethanol as needed to reach a final lipid concentration of 6 mg/mL.
NOTE: This is the same lipid solution composition as in 2.2.1 but with a total volume of 1000 µL. - Dilute the 100 mM pH 4 acetate buffer stock to 10 mM in a total volume of 8 mL as the MIVM effluent quench bath.
NOTE: A magnetic stir bar may also be added to the quench bath.
- Mix the two ethanolic lipid solvent stream solutions in a 1.5 mL microcentrifuge tube, as given in Table 2, by diluting the lipid stock solutions and adding additional ethanol as needed to reach a final lipid concentration of 6 mg/mL.
- Formulation of LNPs using a MIVM mixer
- Place the 8 mL quench bath underneath the mixer stand so that the outflow tubing is emptying into the quench bath.
- Draw up the solvent and antisolvent streams into 1 mL gas-tight syringes using a blunt tip needle. Remove all air bubbles and dispose of the needle. Prime each syringe by inverting and expelling any air from the syringe.
- Assemble the four syringes onto the mixer in a clockwise fashion (Syringes 1-4, Table 2) with the same streams on the opposite sides. See Figure 1B for the final appearance schematic.
- Hold the bearing housing on both sides of the mobile plate. Avoid placing the fingers on the bottom face of the housing, as this poses a pinch hazard due to the mechanical stops. Carefully lower the mobile plate until it evenly rests on the syringes.
NOTE: The syringe plungers must all be at an equal height before operation. - Steadily and smoothly depress the plate, aiming to complete the operation in about 0.5 s to 1 s for these stream volumes.
- Cap the quench bath, which contains the LNP dispersion.
- Perform the cleaning of the MIVM after use, following the instructions in step 3.5 below.
- Formulation of LNPs using a MIVM mixer driven by a syringe pump
- Assemble the MIVM as described in step 3.1.
- Prepare the solvent and antisolvent solutions in the desired composition and at a sufficient volume for the required formulation size (Table 3).
NOTE: These are the same compositions as the aqueous antisolvent (step 1.4) and ethanolic solvent (step 3.2.1) streams but are scaled up for use with the larger syringe volumes in Table 3. - Load the solutions into gas-tight syringes with 20 mL volumes and attach PTFE tubing with a luer adapter fitted on the end. Prime the syringes and tubing by inverting the syringe and tubing and then expelling any air.
- Mount the syringes into a syringe pump and attach the syringes to the mixer inlets on the MIVM, as shown in Figure 3A.
NOTE: The CIJ may also be operated via pumps in the same way but with only one antisolvent and solvent stream syringe. - Dilute the 100 mM pH 4 acetate buffer stock to 10 mM in a total volume of 320 mL as the MIVM effluent quench bath.
- Place the quench bath underneath the MIVM outlet.
- Set the volumetric flow rate on the syringe pump to 20 mL/min.
NOTE: The volumetric flow rates may be varied from 2 mL/min to 40 mL/min by manually setting the values on the syringe pump to form LNPs with different micromixing conditions. - Start the syringe pump, but allow the first 10 s of effluent to flow into a waste beaker. Collect the MIVM effluent in the quench bath after this 10 s start-up flow period.
- Remove and cap the quench bath containing LNP dispersion made at the selected (20 mL/min) volumetric flow rate.
NOTE: This procedure may be repeated to synthesize LNPs at different flow rates by selecting appropriate quench bath volumes. - Clean the MIVM between each experiment (when changing the flow rate) by flushing out at least twice the volume of outlet tubing to prevent collection of LNPs made at the previous flow rate condition before beginning the next LNP synthesis.
- Equipment cleaning after use
- Detach the mixer from the stand while keeping the syringes attached, and hold it over a waste container. Remove the syringes, letting the hold-up volume drain into the container. Then, hold the mixer assembly upside down and use the spanner wrench to disassemble the mixer.
- Rinse the outlet tubing with solvent (e.g., ethanol) and dry with air or nitrogen.
- Rinse the mixing geometry with a suitable solvent, such as deionized water or ethanol, followed by a rinse with ethanol. Dry the components using a stream of air or nitrogen.
- Rinse the O-ring with deionized water and blot dry.
NOTE: If the O-ring looks stretched or deformed, let it air dry overnight before using it. Maintain a large stock of O-rings since they are consumable parts. If the shape does not recover by the next day, dispose of the O-ring. - Rinse the top disk thoroughly with solvent, using an air or nitrogen stream to dry the surface and the syringe fittings.
- Rinse each syringe with a good solvent (e.g., deionized water or ethanol). Apply a final rinse with ethanol and air dry before the next use.
4. Post-processing of LNPs
- Buffer exchange and ethanol removal
- Dialyze the LNP dispersion against a 10 mM HEPES buffer at pH 7.4 utilizing an appropriately sized dialysis cassette with a 20 kDa molecular weight cutoff dialysis cartridge.
NOTE: This step both removes the residual 10% ethanol and increases the dispersion pH by replacing the acetate buffer with HEPES. - Dilute the stock 1 M HEPES buffer to 10 mM in a total volume of 1 L.
- Load the LNP dispersion into a 12 mL dialysis cassette using a syringe and needle.
- Immerse the dialysis cartridge in the 1 L of HEPES buffer and magnetically stir the external buffer. Change the external HEPES buffer after 3 h, and remove the dialysis cartridge after an additional 3 h for a total dialysis time of 6 h.
- Withdraw the dialyzed LNP dispersion from the dialysis cartridge and discard the spent cartridge.
- Dialyze the LNP dispersion against a 10 mM HEPES buffer at pH 7.4 utilizing an appropriately sized dialysis cassette with a 20 kDa molecular weight cutoff dialysis cartridge.
- LNP suspension concentration via ultracentrifugation
- Pipette the suspension into an appropriately sized centrifugal filter with a MWCO of 100kDa30.
NOTE: Centrifugal filters are available in a range of sizes, commonly from 0.5 mL to 12 mL. - Centrifuge the dispersion at 2000 x g for 10-20 min (at room temperature) to increase the concentration by a factor of approximately 5-20x.
NOTE: During centrifugation, check on the sample every 5 min to ensure that an appropriate amount of liquid remains.
- Pipette the suspension into an appropriately sized centrifugal filter with a MWCO of 100kDa30.
5. Characterization of LNPs
- Hydrodynamic diameter measurements with dynamic light scattering (DLS)
- Dispense 750 µL or 900 µL of the prepared LNP dispersion (without any dilutions) into a plastic micro or square cuvette, respectively.
NOTE: Smaller volumes can also be used in appropriate cuvettes. - Set the appropriate viscosity for the solvent in which the LNPs are dispersed (i.e., 1.26 cP for 10 vol% ethanol mixture).
- Collect backscattered light at a 173° angle at 25 °C, followed by the size determination of LNP using the Stokes-Einstein model and the first cumulant of the light scattering correlation function series expansion as defined in the ISO standard document 13321:1996 E.
- Repeat the measurements at least three times.
- Dispense 750 µL or 900 µL of the prepared LNP dispersion (without any dilutions) into a plastic micro or square cuvette, respectively.
- Zeta potential measurements
- Add ~800 µL of as-prepared LNP dispersion into folded capillary zeta-sizer cells.
NOTE: To avoid bubble entrapment within the capillary channel, half of the liquid is dispensed into the cells, held upside down, and then rotated. - Repeat the measurements at least three times.
NOTE: Buffer conductivity should be between 0.2-2 milliSiemens/cm for best results.
- Add ~800 µL of as-prepared LNP dispersion into folded capillary zeta-sizer cells.
- Encapsulation efficiency (EE) measurements by commercially available RNA quantification kit
- Dilute the appropriate amount of the 20x Tris-EDTA (TE) buffer at pH 7.5 provided in the assay kit to 1x concentration using nuclease-free water.
- Prepare a Triton X-100 solution at 2 wt%.
- Dilute the appropriate amount of LNP dispersions in the 1x TE buffer to yield a total mass concentration of approximately 0.6 µg/mL for RNA, with ethanol content below 0.2 vol%.
- Prepare similar samples as in the previous step but containing 0.5 wt.% Triton X-100, which effectively dissolves the LNPs, facilitating the differentiation between "free" and total RNA concentrations in the absence and presence of Triton X-100, respectively.
- Prepare appropriate amounts of control RNA solutions at the concentrations of 0 µg/mL, 0.1 µg/mL, 0.2 µg/mL, 0.4 µg/mL, 0.6 µg/mL, 0.8 µg/mL and 1.0 µg/mL in the 1x TE buffer.
NOTE: The RNA kit may initially need to be diluted. - Repeat the previous step but at 0.5 wt.% Triton X-100.
- Dilute the Ribogreen dye (provided in the kit) 200 times in the 1x TE buffer.
- Add appropriate volumes of the diluted Ribogreen dye to all prepared control RNA and sample solutions, whether with or without Triton X-100. The dye should be diluted equally across both control RNA and sample solutions at a factor of two. Consequently, the total dilution of the dye is 400 times that of its original concentration in the assay kit.
- Vortex mix the containers.
- Prepare a 96-well, non-treated, flat-bottom, opaque plate for analysis in a plate reader.
- Dispense 100 µL volumes of samples and RNA controls into separate cells of the plate.
NOTE: Watch for bubble formation in solutions containing Triton X-100. At least three replicates are conducted for each sample, requiring at least 350 µL of samples. - Record fluorescence intensity using the plate reader at an excitation wavelength of 485 nm and an emission wavelength of 528 nm, with an illumination duration of approximately 10 s per reading. No shaking is needed.
NOTE: The EE is determined from , where Iwith Triton and Iwithout Triton represent the averaged fluorescence intensity of three replicates of samples with and without Triton X-100, respectively, and awith Triton and awithout Triton denote the slope of linear fits to the two averaged calibration curves with and without Triton X-100, respectively31.
, where Iwith Triton and Iwithout Triton represent the averaged fluorescence intensity of three replicates of samples with and without Triton X-100, respectively, and awith Triton and awithout Triton denote the slope of linear fits to the two averaged calibration curves with and without Triton X-100, respectively31.
Representative Results
Screening of optimal LNP formulations can be rapidly achieved with relatively small quantities of material using two-stream CIJ turbulent mixers, provided they are operated at appropriate velocities. A µMIVM mixer driven by a programmable syringe pump, as depicted in Figure 3A, is utilized to highlight the importance of achieving sufficient micromixing, above a critical Reynolds number to form small, monodisperse LNPs. Table 3 contains the summary of formulations used for producing LNPs, and the mixer setup follows the protocol outlined in step 3.4. LNP sizes were characterized by dynamic light scattering (DLS) as a function of Reynolds number. As shown in Figure 3B, beyond a critical Re of 5000, small and monodisperse LNPs are observed. Moreover, high Reynolds numbers (~104-105) can be accessed when the syringes are depressed uniformly by handheld operation or by using the mixing stand (right-most data point in Figure 3B). The mixing stand, shown in Figure 2A, uniformly depresses all syringes simultaneously27. As a result, such LNPs also have optimal sizes. With inadequate mixing (i.e., too small of a Re), larger LNPs are formed. Photos representative of LNPs formed at low (Photo 1) and high Re (Photo 2) are shown in Figure 3B. Samples made at low Re are turbid, indicating the presence of large colloidal light scattering structures (Tyndall effect), but LNPs formed at high Re appear clear due to weaker, blue-shifted scattering from smaller colloids.
Ionizable lipids have different physical-chemical properties, which affect the physicochemical properties of LNPs produced with otherwise identical lipid formulations. A CIJ mixer (Figure 1A) is used to test this. Table 4 lists the formulations made using two FDA-approved ionizable lipids: ALC-0315 and MC3. Figure 4A shows that the LNPs made at pH 5 from ALC-0315 are ~80 nm, whereas LNPs made using MC3 are ~60 nm. Moreover, at pH 5, MC3-LNPs have a positive zeta potential (~28 mV), while the ALC-LNPs have a neutral zeta potential (<10 mV). This distinction in surface charge as well as the overall size of LNPs, arises from the pKa of the two lipids. MC3 has a higher pKa (6.44) when compared to ALC0315 (6.09)32; therefore, a higher fraction of MC3 lipids are charged at pH 5. Both formulations have 1.5 mol% PEG-lipid stabilizer; however, the MC3-LNPs stabilize at a smaller size due to larger electrostatic repulsions during LNP assembly during the diffusion-limited aggregation, which arrests growth at the smaller size. Both the formulations show high encapsulation efficiencies (>90%), as shown in Figure 4B. The chemistry of lipids is crucial in determining the overall properties as well as the performance of the LNPs, and thus, they must be chosen carefully based on the target application.
LNPs made using both of the turbulent mixer geometries (step 2.3 and step 3.3) have similar physicochemical properties. This comparison is further extended to LNPs made from poor mixing techniques, such as bulk pipette mixing, to further illustrate the distinction between turbulent mixers and non-uniform mixing techniques (Figure 1C). Table 5 provides the summary of formulations used for making LNPs, as shown in Figure 5. In the case of the pipette mixing technique, equal volumes of ethanol and aqueous streams are rapidly mixed by pipetting up and down for 15-20 s, followed by pipetting the mixture into an acetate buffer bath at pH 4. Figure 5A shows that the sizes of LNPs in the quench 10 mM acetate buffer bath (pH 4, 10 vol% ethanol) are strikingly similar and small (~50 nm) irrespective of the mixer geometry used (CIJ or MIVM). However, LNPs made using pipette mixing are twice the size of LNPs made using turbulent mixers. This shows that the LNPs made from different CIJ geometries exhibit similar properties when made at sufficiently high velocities (turbulent regime above the critical Reynolds number), while poor mixing results in larger, polydisperse LNPs.
Next, the LNPs are dialyzed against a 10 mM HEPES buffer, pH 7.4 (100x volume), to remove ethanol and switch the pH to 7.4. During this process, there is some LNP fusion and growth to a slightly larger size, as shown in Figure 5A, which is in line with the well-studied fusion mechanism in the literature33. Overall, the LNPs made using CIJ and MIVM mixers are less than 100 nm, while the LNPs made using pipette mixing are around 140 nm. As shown in Figure 5B, the zeta potentials for these formulations are less than 10 mV, indicating that they are all neutral at pH 7.4. Additionally, they all show high encapsulation efficiencies of >95% (Figure 5C). Thus, the LNPs with optimal physicochemical properties can be easily manufactured using turbulent mixer technologies. The performance of these LNPs is assessed by carrying out in vitro transfection in HeLa cells.
Luciferase-based in vitro transfection assay protocol is adopted from a previous publication34. Figure 6A plots the luminescence (RLU) per 1000 cells for the three formulations summarized in Table 5. Lipofectamine 3000 is used as a positive control. It is important to note that lipofectamine 3000 is generally used for DNA formulations; however, it worked as an adequate control in these experiments. LNPs made using 2-jet CIJ and 4-jet MIVM mixers transfect much better than the LNPs made using pipette mixing. Even though the larger particles are expected to transfect better than the small particles due to the greater payload per LNP, the LNPs made using pipette mixing here transfect less effectively. This clearly implies that there is a significant difference in the structures of LNPs made with CIJ technologies compared to the LNPs made with pipette mixing. The transfection efficiencies of LNPs made with CIJ and MIVM mixers are essentially identical. Lipofectamine 3000 shows the lowest transfection efficiency. Figure 6B evaluates the LNP toxicity in vitro on HeLa cells using a cell viability assay based on sodium resazurin salt35. All the formulations show low cytotoxicity, exhibited by high levels of cell viability when plotted as a percentage of cells alive versus the control that does not have any nanoparticle treatment.

Figure 1: Mixing methods for producing LNPs. (A) The two-jet confined impinging jet mixer (CIJ) showing a photograph of a transparent mixer and an assembled Delrin mixer setup regularly used in the laboratory. (B) The four-jet micro multiple inlet vortex mixer (µMIVM) showing a photograph of a transparent mixer and an assembled stainless steel and Delrin mixer setup. The inlet streams for the transparent mixer have been moved to the sides of the mixer for better visualization of the mixing geometry, while in the practical mixer, the inlet streams enter from the top. Both the CIJ and µMIVM operate with sufficient liquid velocity that flows are in the turbulent regime, and mixing produces Kolmogorov microscales smaller than 1 µm, which enables the attainment of supersaturation in ~1.5 ms. (C) The pipette mixing setup widely used to prepare small volumes of LNP dispersions by mixing aqueous and ethanolic solutions. Please click here to view a larger version of this figure.

Figure 2: Expanded view of the µMIVM and its mixing stand. (A) Assembled µMIVM, with glass syringes, under the mixer stand that facilitates even and rapid depression of the syringes. This figure is reproduced from Markwalter et al.29. (B) Disassembled µMIVM showing the interior components. This mixing geometry is identical to the transparent mixer in Figure 1B, except that the inlet streams enter through the top disk and not through the lateral cylindrical surface. Please click here to view a larger version of this figure.
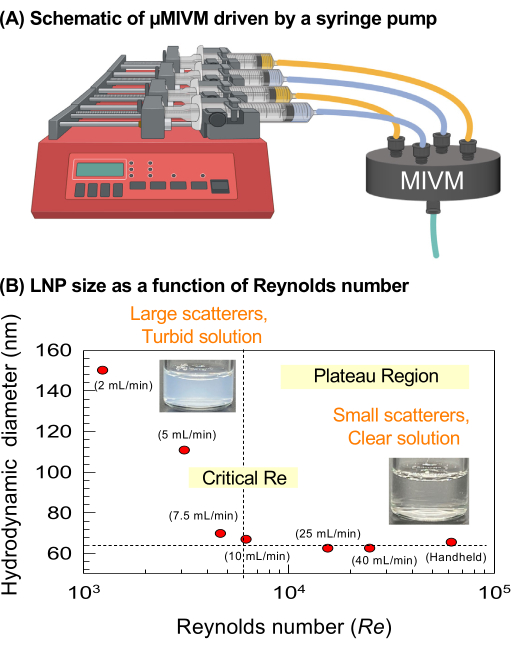
Figure 3: Increasing Reynolds number in a turbulent mixer decreases LNP hydrodynamic diameter up to a plateau critical Reynolds number. (A) Schematic depiction of the syringe pump and µMIVM setup used to control the Reynolds number in the turbulent mixer by setting stream volumetric flow rates. (B) LNP hydrodynamic diameter versus Reynolds number in the MIVM. Increasing the Reynolds number and turbulent energy dissipation improves mixing and leads to more homogeneous mixing, supersaturation, and growth of particles. Above the critical Reynolds number, LNP sizes remain constant with increasing flow rates due to the Da<<1 condition, i.e., the solvent/antisolvent diffusion time is shorter than the NP assembly time. The flow rates given are the total flow rates of all the four streams. Please click here to view a larger version of this figure.

Figure 4: Colloidal properties of LNPs produced with different ionizable lipids. (A) Hydrodynamic diameters and zeta potentials of LNPs produced with two different ionizable lipids: ALC-0315 and MC3. Measurements are made at pH 5 in the quench bath after LNP formation. Differences in apparent pKa of the ionizable lipids affect the colloidal properties of LNPs. (B) Encapsulation efficiency measurements of both the LNPs (n = 3, error bars represent one standard deviation). Please click here to view a larger version of this figure.

Figure 5: Colloidal properties of LNPs produced with different mixers encapsulating luciferase mRNA using MC3 ionizable lipid. (A) Hydrodynamic diameters of LNPs produced with 2-jet, 4-jet, and pipette mixers. Measurements are performed both under the acidic conditions of the quench bath after LNP formation (left-hand side) and after dialysis into a neutral HEPES buffer (right-hand side). LNP size grows during pH neutralization due to deionization of the ionizable lipid, leading to LNP fusion and growth. The lipid-PEG stabilizer arrests this particle coalescence growth before the formation of micron-sized precipitates. (B) Surface charge measurements (as the ζ-potential) on dialyzed LNPs in the 10 mM HEPES, pH 7.4 condition. All LNPs are within 2 mV of 0 mV, which indicates that these particle surfaces are neutral and have only a nearly undetectable amount of cationic charge. (C) Encapsulation efficiency measurements after dialysis of LNPs (n = 3, error bars represent one standard deviation). Please click here to view a larger version of this figure.

Figure 6: Transfection of HeLa cells with as-prepared LNPs. (A) Luminescence of expressed luciferase enzyme after treatment with luciferin. (B) Viability of HeLa cells after incubation with LNPs. Cells show no statistically significant change in viability, as indicated by a resazurin metabolic assay (n = 4, error bars represent one standard deviation). Please click here to view a larger version of this figure.
| Stream(s) / Quench bath |
Component | Formulation | Stock | Volume |
| Syringe 1 – Ethanol stream (6 mg/mL total lipid) |
Ionizable lipid (MC3) | 50 mol% | 50 mg/mL | 0.5 mL |
| Zwitterion lipid (DSPC) | 10 mol% | 5 mg/mL | ||
| Cholesterol | 38.5 mol% | 5 mg/mL | ||
| Pegylated lipid (DMG-PEG2000) | 1.5 mol% | 4 mg/mL | ||
| Syringe 2 – Aqueous stream (0.3 mg/mL RNA) |
Yeast RNA | N/P 6 | 10 mg/mL | 0.5 mL |
| Acetate Buffer | 20 mM, pH 4 | 100 mM, pH 4 | ||
| Quench bath | Acetate Buffer | 10 mM, pH 4 | 100 mM, pH 4 | 4 mL |
Table 1: Standard patisiran LNP Formulation produced by a CIJ mixer. Yeast RNA is used as a model RNA for this protocol. All the solutions are molecularly dissolved and mixed thoroughly before loading them in the syringes.
| Stream(s) / Quench bath |
Component | Formulation | Stock | Volume |
| Syringe 1 – Ethanol stream (6 mg/mL total lipid) |
Ionizable lipid (MC3) | 50 mol% | 50 mg/mL | 0.5 mL |
| Zwitterion lipid (DSPC) | 10 mol% | 5 mg/mL | ||
| Cholesterol | 38.5 mol% | 5 mg/mL | ||
| Pegylated lipid (DMG-PEG2000) | 1.5 mol% | 4 mg/mL | ||
| Syringe 2 – Aqueous stream (0.3 mg/mL RNA) |
Yeast RNA | N/P 6 | 10 mg/mL | 0.5 mL |
| Acetate Buffer | 20 mM, pH 4 | 100 mM, pH 4 | ||
| Syringe 3 – Ethanol stream (6 mg/mL total lipid) |
Ionizable lipid (MC3) | 50 mol% | 50 mg/mL | 0.5 mL |
| Zwitterion lipid (DSPC) | 10 mol% | 5 mg/mL | ||
| Cholesterol | 38.5 mol% | 5 mg/mL | ||
| Pegylated lipid (DMG-PEG2000) | 1.5 mol% | 4 mg/mL | ||
| Syringe 4 – Aqueous stream (0.3 mg/mL RNA) |
Yeast RNA | N/P 6 | 10 mg/mL | 0.5 mL |
| Acetate Buffer | 20 mM, pH 4 | 100 mM, pH 4 | ||
| Quench bath | Acetate Buffer | 10 mM, pH 4 | 100 mM, pH 4 | 8 mL |
Table 2: Standard patisiran LNP Formulation produced by an MIVM mixer. An MIVM mixer utilizes four streams – two solvents and two antisolvents. Streams with unequal momenta may be used; however, streams with equal momenta are chosen for this protocol.
| Stream(s) / Quench bath |
Component | Formulation | Stock | Volume |
| Syringe 1 – Ethanol stream (6 mg/mL total lipid) |
Ionizable lipid (MC3) | 50 mol% | 50 mg/mL | 20 mL |
| Zwitterion lipid (DSPC) | 10 mol% | 5 mg/mL | ||
| Cholesterol | 38.5 mol% | 5 mg/mL | ||
| Pegylated lipid (DMG-PEG2000) | 1.5 mol% | 4 mg/mL | ||
| Syringe 2 – Aqueous stream (0.3 mg/mL RNA) |
Yeast RNA | N/P 6 | 10 mg/mL | 20 mL |
| Acetate Buffer | 20 mM, pH 4 | 100 mM, pH 4 | ||
| Syringe 3 – Ethanol stream (6 mg/mL total lipid) |
Ionizable lipid (MC3) | 50 mol% | 50 mg/mL | 20 mL |
| Zwitterion lipid (DSPC) | 10 mol% | 5 mg/mL | ||
| Cholesterol | 38.5 mol% | 5 mg/mL | ||
| Pegylated lipid (DMG-PEG2000) | 1.5 mol% | 4 mg/mL | ||
| Syringe 4 – Aqueous stream (0.3 mg/mL RNA) |
Yeast RNA | N/P 6 | 10 mg/mL | 20 mL |
| Acetate Buffer | 20 mM, pH 4 | 100 mM, pH 4 | ||
| Quench bath (Collection time = 30 s) |
HEPES Buffer | 10 mM, pH 7.5 | 1 M, pH 7.5 | 320 mL |
Table 3: LNP Formulation produced by a MIVM mixer driven by a syringe pump. DODMA is used as an ionizable lipid along with yeast RNA as the model RNA. An example run with 40 mL/min is chosen for this protocol.
| Stream(s) / Quench bath |
Component | Formulation | Stock | Volume |
| Syringe 1 – Ethanol stream (12 mg/mL total lipid) |
Ionizable lipid (MC3 or ALC0315) | 50 mol% | 50 mg/mL | 0.5 mL |
| Zwitterion lipid (DSPC) | 10 mol% | 5 mg/mL | ||
| Cholesterol | 38.5 mol% | 5 mg/mL | ||
| Pegylated lipid (DMG-PEG2000) | 1.5 mol% | 4 mg/mL | ||
| Syringe 2 – Aqueous stream (0.6 mg/mL RNA) |
Yeast RNA | N/P 6 | 10 mg/mL | 0.5 mL |
| Acetate Buffer | 20 mM, pH 5 | 100 mM, pH 5 | ||
| Quench bath | Acetate Buffer | 10 mM, pH 5 | 100 mM, pH 5 | 9 mL |
Table 4: LNP Formulations from two different ionizable lipids produced by a CIJ mixer. Formulations are made from either ALC-0315 or MC3, while all other components are kept the same.
| Stream(s) / Quench bath |
Component | Formulation | Stock | Volume |
| Syringe 1 – Ethanol stream (6 mg/mL total lipid) |
Ionizable lipid (MC3) | 50 mol% | 50 mg/mL | 0.5 mL |
| Zwitterion lipid (DSPC) | 10 mol% | 5 mg/mL | ||
| Cholesterol | 38.5 mol% | 5 mg/mL | ||
| Pegylated lipid (DMG-PEG2000) | 1.5 mol% | 4 mg/mL | ||
| Syringe 2 – Aqueous stream (0.3 mg/mL RNA) |
FLuc mRNA | N/P 6 | 1 mg/mL | 0.5 mL |
| Acetate Buffer | 20 mM, pH 4 | 100 mM, pH 4 | ||
| Quench bath | Acetate Buffer | 10 mM, pH 4 | 100 mM, pH 4 | 4 mL |
Table 5: Standard patisiran LNP Formulation produced by a CIJ mixer. FLuc mRNA that expresses a luciferase protein is employed to measure gene expression using a bioluminescence assay.
Supplementary File 1: Suppliers of CIJ and MIVM Mixers. Please click here to download this File.
Discussion
Synthesis of LNPs containing nucleic acid polymers using two confined impinging jet turbulent mixers has been presented. When conducted at appropriate velocities, CIJ turbulent mixers ensure the time scale of mixing is shorter than the LNP assembly time, producing homogenous supersaturation conditions for forming small LNPs with narrow size distributions21. Consequently, LNPs made with the same chemistry using different turbulent mixer geometries (the 2-jet CIJ and the 4-jet MIVM mixer) exhibit similar physicochemical properties and show good transfection efficiencies (Figure 5 and Figure 6). In contrast, LNPs made using pipetting that produces poorer mixing results in larger and more polydisperse LNPs (Figure 5A) with lower transfection efficiencies. It has long been understood that mixing and assembly kinetics play a significant role in LNP processing; Cullis et al. noted that rapid convective-diffusive mixing of ethanol and buffer leads to the formation of small particles with a narrow size distribution, whereas slow diffusive mixing leads to larger particles with broad size distributions9. The time scale of mixing in CIJ turbulent mixers decreases proportionately to the inlet flow rates of the streams to the mixer27. This is quantified by the dimensionless Reynolds number (Re), which measures the ratio between the inertial and viscous forces. The turbulence inside the mixing chambers of the CIJ and MIVM occurs at sufficiently high Re, such that the turbulent vortex stretching results in small scales of length that produce rapid solvent/antisolvent mixing by diffusion. The turbulent length scale depends on the Re and not the specific geometry of the mixing device. That is why either the CIJ or the MIVM makes the same LNP particles, and why various sizes of MIVM mixers make the same NP sizes27. At high Re, corresponding to high inlet velocities, LNPs can be made reproducibly without batch-to-batch variations (Figure 3B).
This protocol enables the formulation of a variety of mRNA, DNA, or siRNA LNPs with different physicochemical properties using turbulent CIJ mixers. In addition to allowing versatility in composition and concentrations, this technique provides a clear path to rapidly screen formulations at bench sale (a few milligrams) and scale up the lead formulations to larger industrial batch sizes at production rates of 5 L/min36. This has been a major hurdle for several other techniques, including bulk-mixing and microfluidics. For instance, bulk-processing techniques fail to consistently manufacture LNPs reproducibly, even at a few milliliter scales. Microfluidic techniques provide a significant improvement over bulk-mixing techniques to enable the production of uniform and reproducible LNPs; however, they are only in the milligram range29. As detailed in the introduction, parallelization of microfluidic devices provides an attempt at scaling to production scales but does not eliminate the issue of fouling, and it cannot be scaled as successfully as mixers based on confined impinging jet technology.
Apart from these advantages, CIJ mixers will be instrumental in manufacturing next-generation LNPs that exhibit targeting capabilities or perform gene editing. The current LNP formulations have lipids and nucleic acids that have similar diffusivities, and therefore, they can be made even with slightly poor mixing at bench-scale. However, gene-editing approaches may require the encapsulation of nucleic acid species with widely different molecular weights, such as small guide RNA molecules and large mRNA transcripts, to code a CAS9 protein37. The very different diffusion time scales of these different species make uniform encapsulation at stoichiometric ratios challenging. This problem of uniform encapsulation becomes more pronounced as the mixing efficiency becomes poorer. Likewise, targeting non-hepatic cells might need the incorporation of strongly bound slow-diffusing stabilizers (such as large molecular weight block copolymers with targeting ligands). Targeting ligands as large as 14 kDa can be conjugated to block copolymers prior to nanoparticle assembly, which enables their uniform incorporation into NPs using CIJ mixing38. CIJ turbulent mixers are useful tools for manufacturing LNPs made with components having different diffusivities.
While CIJ turbulent mixers demonstrate several advantages over other mixers for formulating LNPs, it is important to note the limitations associated with each geometry. The 2-jet CIJ mixer requires that both the inlet streams (ethanol and water) have equal momenta (within 10%-30%) to achieve uniform turbulent micromixing in the chamber. The fact that the exit stream comprises 50:50 solvent/antisolvent limits the level of supersaturation in the mixing cavity where precipitation occurs29. This drawback is addressed by the 4-jet MIVM mixer, as it can utilize four jets with unequal momenta to accomplish high supersaturation conditions in the mixing chamber. Additionally, both the mixers are required to be in the order of milligrams of total mass, making them a non-ideal choice for high-throughput screening of many different LNP formulations. For simple LNP formulations, screening may best be done with microfluidics or pipetting strategies at microgram scales and then transferred to the confined impinging jet technology when a few lead formulations have been identified. It is also crucial to consider the dead volumes in the mixers. In the CIJ, two jet mixers, the hold-up volumes are 50-100 microliters. This amount of material must be subtracted from the amount captured in the quench bath when calculating the recovery from the process. These losses are insignificant when operating on large scales but would account for 10% losses when total volumes of 5 mL are produced, as shown here. The impinging jet turbulent mixers are a valuable tool for producing LNPs at the GMP scale, as evidenced by the two FDA-approved COVID-19 vaccines.
Disclosures
The authors have nothing to disclose.
Acknowledgements
NSF Fellowship to BKW (DGA1148900), support from Tessera Therapeutics Inc., the Bill and Melinda Gates Foundation (BMGF, contract numbers OPP1150755 and INV-041182), and the FDA under award 75F40122C00186.
Materials
| 18:0 PC (DSPC) | Avanti Polar Lipids | 850365P | Helper lipid |
| 21 G x 1-1/2 in. BD PrecisionGlide Needle | BD | 305167 | |
| 96 Well Black Wall Black Bottom Plate | Fisher Scientific | 07-000-135 | |
| 96 Well White/Clear Bottom Plate, TC Surface | Thermo Fisher Scientific | 165306 | |
| Acetic Acid, Glacial | Fisher Scientific | A38-212 | |
| ALC-0315 | Avanti Polar Lipids | 890900 | Ionizable lipid |
| Amicon Ultra Centrifugal Filter, 100 kDa MWCO, 15 mL | Millipore Sigma | UFC910024 | |
| Amicon Ultra Centrifugal Filter, 100 kDa MWCO, 4 mL | Millipore Sigma | UFC810096 | |
| Bright-Glo Luciferase Assay System | Promega | E2620 | |
| Cholesterol | Millipore Sigma | C8667 | |
| CleanCap FLuc mRNA (5 moU) | Trilink Biotechnologies | L-7202 | |
| Confined Impinging Jets Mixer | Holland Applied Technologies, Helix Biotech, Diamond Tool and Die (DTD) | N/A | Contact Holland or DTD for custom orders and the Helix Biotech system is Nova BT. Review text for new mixer validation |
| D-Lin-MC3-DMA | MedChemExpress | HY-112251 | Ionizable lipid |
| DMEM, high glucose, pyruvate | Thermo Fisher Scientific | 11995065 | |
| DMG-PEG 2000 | Avanti Polar Lipids | 880151P | PEG-lipid |
| DODMA | Avanti Polar Lipids | 890899P | Ionizable lipid |
| Ethanol 200 Proof | Decon Labs, Inc. | 2701 | |
| Falcon 15 mL Conical Centrifuge Tubes | Fisher Scientific | 14-959-70C | |
| Falcon 50 mL High Clarity Conical Centrifuge Tubes | Fisher Scientific | 14-959-49A | |
| Fetal Bovine Serum, certified, United States | Thermo Fisher Scientific | 16000044 | |
| HeLa | ATCC | CCL-2 | |
| HEPES, free acid | IBI Scientific | IB01130 | |
| HSW HENKE-JECT two-part 1 mL Luer | Henke Sass Wolf | 4010.200V0 | |
| HSW HENKE-JECT two-part 5 mL (6 mL) Luer Lock | Henke Sass Wolf | 4050.X00V0 | |
Idex 1648 ETFE tubing  ” OD 0.093” ID ” OD 0.093” ID |
Idex Health & Science | 1648 | |
| Idex P-678 ¼”-28 to Luer fitting | Idex Health & Science | P-678 | |
| Idex P-940 ferrule for ETFE tubing | Idex Health & Science | P-940 | |
| Lipofectamine 3000 Transfection Reagent | Thermo Fisher Scientific | L3000001 | |
| Luer fitting | Idex Health & Science | P-604 | Assemble on CIJ or MIVM mixer inlet with corresponding threads. Idex parts are also available through VWR and many other suppliers |
| Mixer stand | Holland Applied Technologies | N/A | See Markwalter & Prud'homme for design.26 Contact Holland for Purchase |
| Multi-Inlet Vortex Mixer | Holland Applied Technologies and Diamond Tool and Die (DTD) | N/A | Contact Holland or DTD for custom orders. Review text for new mixer validation |
| O-ring (MIVM) | C.E. Conover | MM1.5 35.50 V75 | Order bulk – consumable part. Ensure solvent compatibility if using an alternative source. |
| Outlet ferrule – CIJ | Idex Health & Science | P-200 | Assemble with outlet fitting (large end flush with tubing) |
| Outlet fitting – CIJ | Idex Health & Science | P-205 | Assemble with ferrule and tubing on CIJ chamber outlet |
| Outlet fitting – MIVM | Idex Health & Science | P-942 | Combination with ferrule |
| Outlet tubing – CIJ | Idex Health & Science | 1517 | Use a tubing cutter for clean ends. Ensure extra tubing doesn't protrude into mixing chamber |
| Outlet tubing – MIVM | N/A | N/A | Fit to ferrule ID. |
| PBS – Phosphate-Buffered Saline (10x) pH 7.4, RNase-free | Thermo Fisher Scientific | AM9624 | |
| Penicillin-Streptomycin (10,000 U/mL) | Thermo Fisher Scientific | 15140122 | |
| PHD 2000 Programmable Syringe Pump | Harvard Apparatus | N/A | |
| Plastic two-piece syringe 1 mL | Thermo Fisher Scientific | S7510-1 | |
| Plug fitting | Idex Health & Science | P-309 | Assemble on CIJ mixer sides (seal access point from drilling) |
| Quant-it RiboGreen RNA Assay Kit and RiboGreen RNA Reagent, RediPlate 96 RiboGreen RNA Quantitation Kit | Invitrogen by Thermo Fisher Scientific | R11491 | |
| Resazurin, Sodium Salt | Thermo Fisher Scientific | R12204 | |
| RNase AWAY Surface Decontaminant | Thermo Fisher Scientific | 7000TS1 | |
| Scintillation vial | DWK Lifesciences | 74504-20 | |
| SGE Gas Tight Syringes, Luer Lock, 100 mL | SGE | 100MR-LL-GT | |
| SGE Gas Tight Syringes, Luer Lock, 50 mL | SGE | 50MR-LL-GT | |
| Slide-A-Lyzer Dialysis Cassettes, 20 K MWCO | Thermo Fisher Scientific | 66012 | |
| Sodium Acetate | Millipore Sigma | 32319-500G-R | |
| Sodium Hydroxide | Fisher Scientific | S320-500 | |
| Sucrose | Millipore Sigma | S7903-1KG | |
| Syringe Filters, Sterile | Genesse Scientific | 25-243 | |
| Triton X-100 | Millipore Sigma | 9036-19-5 | |
| Trypsin-EDTA (0.25%), phenol red | Thermo Fisher Scientific | 25200056 | |
| Water, Endotoxin Free | Quality Biological | 118-325-131 | RNAse and DNAse free |
| Yeast RNA (10 mg/mL) | Thermo Fisher Scientific | AM7118 |
References
- Hou, X., Zaks, T., Langer, R., Dong, Y. Lipid nanoparticles for mRNA delivery. Nat Rev Mater. 6 (12), 1078-1094 (2021).
- Dowdy, S. F., Setten, R. L., Cui, X. -. S., Jadhav, S. G. Delivery of RNA therapeutics: The great endosomal escape. Nucleic Acid Ther. 32 (5), 361-368 (2022).
- Eygeris, Y., Patel, S., Jozic, A., Sahay, G. Deconvoluting lipid nanoparticle structure for messenger RNA delivery. Nano Lett. 20 (6), 4543-4549 (2020).
- Tenchov, R., Bird, R., Curtze, A. E., Zhou, Q. Lipid nanoparticles─from liposomes to mRNA vaccine delivery, a landscape of research diversity and advancement. ACS Nano. 15 (11), 16982-17015 (2021).
- Buck, J., Grossen, P., Cullis, P. R., Huwyler, J., Witzigmann, D. Lipid-based DNA therapeutics: Hallmarks of non-viral gene delivery. ACS Nano. 13 (4), 3754-3782 (2019).
- Semple, S. C., et al. Rational design of cationic lipids for siRNA delivery. Nat Biotechnol. 28 (2), 172-176 (2010).
- Lam, K., et al. Unsaturated, trialkyl ionizable lipids are versatile lipid-nanoparticle components for therapeutic and vaccine applications. Adv Mater. 35 (15), 2209624 (2023).
- Gilleron, J., et al. Image-based analysis of lipid nanoparticle-mediated siRNA delivery, intracellular trafficking and endosomal escape. Nat Biotechnol. 31 (7), 638-646 (2013).
- Evers, M. J. W., et al. State-of-the-art design and rapid-mixing production techniques of lipid nanoparticles for nucleic acid delivery. Small Methods. 2 (9), 1700375 (2018).
- Kulkarni, J. A., Cullis, P. R., Van Der Meel, R. Lipid nanoparticles enabling gene therapies: From concepts to clinical utility. Nucleic Acid Ther. 28 (3), 146-157 (2018).
- Jürgens, D. C., et al. Lab-scale siRNA and mRNA LNP manufacturing by various microfluidic mixing techniques – an evaluation of particle properties and efficiency. OpenNano. 12, 100161 (2023).
- Wang, X., et al. Preparation of selective organ-targeting (sort) lipid nanoparticles (LNPs) using multiple technical methods for tissue-specific mRNA delivery. Nat Protoc. 18 (1), 265-291 (2023).
- Yanez Arteta, M., et al. Successful reprogramming of cellular protein production through mRNA delivered by functionalized lipid nanoparticles. Proc Natl Acad Sci. 115 (15), E3351-E3360 (2018).
- Eygeris, Y., Gupta, M., Kim, J., Sahay, G. Chemistry of lipid nanoparticles for RNA delivery. Acc Chem Res. 55 (1), 2-12 (2022).
- Shepherd, S. J., et al. Scalable mRNA and siRNA lipid nanoparticle production using a parallelized microfluidic device. Nano Lett. 21 (13), 5671-5680 (2021).
- Leung, A. K. K., Tam, Y. Y. C., Chen, S., Hafez, I. M., Cullis, P. R. Microfluidic mixing: A general method for encapsulating macromolecules in lipid nanoparticle systems. J Phys Chem B. 119 (28), 8698-8706 (2015).
- Prakash, G., et al. Microfluidic fabrication of lipid nanoparticles for the delivery of nucleic acids. Adv Drug Deliv Rev. 184, 114197 (2022).
- Carvalho, B. G., Ceccato, B. T., Michelon, M., Han, S. W., De La Torre, L. G. Advanced microfluidic technologies for lipid nano-microsystems from synthesis to biological application. Pharmaceutics. 14 (1), 141 (2022).
- Webb, C., et al. Using microfluidics for scalable manufacturing of nanomedicines from bench to GMP: A case study using protein-loaded liposomes. Int J Pharm. 582, 119266 (2020).
- Warne, N., et al. Delivering 3 billion doses of comirnaty in 2021. Nat Biotechnol. 41 (2), 183-188 (2023).
- Johnson, B. K., Prud’homme, R. K. Flash nanoprecipitation of organic actives and block copolymers using a confined impinging jets mixer. Aust J Chem. 56 (10), 1021-1024 (2003).
- Hogarth, C., et al. Evaluating the impact of systematic hydrophobic modification of model drugs on the control, stability and loading of lipid-based nanoparticles. J Mater Chem B. 9 (48), 9874-9884 (2021).
- Belliveau, N. M., et al. Microfluidic synthesis of highly potent limit-size lipid nanoparticles for in vivo delivery of siRNA. Mol Ther Nucleic Acids. 1, e37 (2012).
- D’addio, S. M., Prud’homme, R. K. Controlling drug nanoparticle formation by rapid precipitation. Adv Drug Deliv Rev. 63 (6), 417-426 (2011).
- Johnson, B. K., Prud’homme, R. K. Mechanism for rapid self-assembly of block copolymer nanoparticles. Phys Rev Lett. 91 (11), 118302 (2003).
- Han, J., et al. A simple confined impingement jets mixer for flash nanoprecipitation. J Pharm Sci. 101 (10), 4018-4023 (2012).
- Markwalter, C. E., Prud’homme, R. K. Design of a small-scale multi-inlet vortex mixer for scalable nanoparticle production and application to the encapsulation of biologics by inverse flash nanoprecipitation. J Pharm Sci. 107 (9), 2465-2471 (2018).
- Johnson, B. K., Prud’homme, R. K. Chemical processing and micromixing in confined impinging jets. AIChE J. 49, 2264-2282 (2003).
- Markwalter, C. E., Pagels, R. F., Wilson, B. K., Ristroph, K. D., Prud’homme, R. K. Flash nanoprecipitation for the encapsulation of hydrophobic and hydrophilic compounds in polymeric nanoparticles. J Vis Exp. (143), e58757 (2019).
- Bailey-Hytholt, C. M., Ghosh, P., Dugas, J., Zarraga, I. E., Bandekar, A. Formulating and characterizing lipid nanoparticles for gene delivery using a microfluidic mixing platform. J Vis Exp. 168, e62226 (2021).
- Bizmark, N., et al. Ribogreen fluorescent assay kinetics to measure ribonucleic acid loading into lipid nanoparticle carriers. Adv Mater Interfaces. 11 (17), 2301083 (2024).
- Zhang, C., et al. Modification of lipid-based nanoparticles: An efficient delivery system for nucleic acid-based immunotherapy. Molecules. 27 (6), 1943 (2022).
- Kulkarni, J. A., et al. Fusion-dependent formation of lipid nanoparticles containing macromolecular payloads. Nanoscale. 11 (18), 9023-9031 (2019).
- El-Mayta, R., Padilla, M. S., Billingsley, M. M., Han, X., Mitchell, M. J. Testing the in vitro and in vivo efficiency of mRNA-lipid nanoparticles formulated by microfluidic mixing. J Vis Exp. (191), e64810 (2023).
- Scalzo, S., et al. Ionizable lipid nanoparticle-mediated delivery of plasmid DNA in cardiomyocytes. Int J Nanomed. 17, 2865-2881 (2022).
- Feng, J., Markwalter, C. E., Tian, C., Armstrong, M., Prud’homme, R. K. Translational formulation of nanoparticle therapeutics from laboratory discovery to clinical scale. J Transl Med. 17 (1), 200 (2019).
- Kazemian, P., et al. Lipid-nanoparticle-based delivery of CRISPR/cas9 genome-editing components. Mol Pharm. 19 (6), 1669-1686 (2022).
- Pinkerton, N. M. . Polymeric drug delivery vehicles and imaging agents. , (2014).

