Embryo Microinjection and Knockout Mutant Identification of CRISPR/Cas9 Genome-Edited Helicoverpa Armigera (Hübner)
Summary
Presented here is a protocol of Helicoverpa armigera (Hübner) embryo microinjection and knockout mutant identification created by CRISPR/Cas9 genome editing. Mutant insects enable further research of gene function and interaction among different genes in vivo.
Abstract
The cotton bollworm, Helicoverpa armigera, is one of the most destructive pests in the world. A combination of molecular genetics, physiology, functional genomics, and behavioral studies has made H. armigera a model species in Lepidoptera Noctuidae. To study the in vivo functions of and interactions between different genes, clustered regularly interspaced short palindromic repeats (CRISPR)/ associated protein 9 (Cas9) genome editing technology is a convenient and effective method used for performing functional genomic studies. In this study, we provide a step-by-step systematic method to complete gene knockout in H. armigera using the CRISPR/Cas9 system. The design and synthesis of guide RNA (gRNA) are described in detail. Then, the subsequent steps consisting of gene-specific primer design for guide RNA (gRNA) creation, embryo collection, microinjection, insect rearing, and mutant detection are summarized. Finally, troubleshooting advice and notes are provided to improve the efficiency of gene editing. Our method will serve as a reference for the application of CRISPR/Cas9 genome editing in H. armigera as well as other Lepidopteran moths.
Introduction
The application of genome editing technology provides an efficient tool to achieve target-gene mutants in diverse species. The emergence of the clustered regularly interspaced short palindromic repeats (CRISPR)/associated protein 9 (Cas9) system provides a novel method to manipulate genomes1. The CRISPR/Cas9 system consists of a guide RNA (gRNA) and the Cas9 endonuclease2,3, while the gRNA can be further divided into two parts, a target complementary CRISPR RNA (crRNA) and a trans-activating crRNA (tracrRNA). The gRNA integrates with Cas9 endonuclease and forms a ribonucleoprotein (RNP). With the gRNA, Cas9 endonuclease can be directed to a specific site of the genome via base complementation. The RuvC and HNH domains of the Cas9 cleave the target site of the genome three bases before the protospacer-adjacent motif (PAM) sequence and create a double-strand break (DSB). The DNA cleavage can then be repaired through two mechanisms, non-homologous end joining (NHEJ) or homology-directed repair (HDR)4. Repair of the DSB introduces insertions or deletions as a way to inactivate the targeted gene, potentially causing a complete loss of gene function. Hence, the hereditable and specificity of the CRISPR/Cas9 system make it a robust method to characterize gene functions in vivo and analyze gene interactions5.
With numerous merits, the CRISPR/Cas9 system has been applied to various fields, including biomedicine6,7, gene therapy8,9, and agriculture10,11,12, and has been used for various biological systems including microorganisms13, plants14,15, nematodes16 , and mammals17. In invertebrates, many insect species have been subjected to CRISPR/Cas9 genome editing, such as the fruit fly Drosophila melanogaster and beyond18,19,20,21,22.
Helicoverpa armigera is one of the most destructive pests worldwide23, and damages numerous crops, including cotton, soybean, and sorghum24,25. With the development of sequencing technology, the genome of H. armigera, as well as that of a range of Lepidoptera insect species, have been sequenced completely26,27,28,29. A large number of resistance and olfactory receptor genes have been identified and characterized from these insects in recent years19,27,28,29. Some resistance-related genes have been identified in H. armigera, such as the genes encoding for cadherin30, an ATP-binding cassette transporter31,32, as well as HaTSPAN133. Knockout of these genes using CRISPR/Cas9 technology results in a high level of resistance to Bacillus thuringiensis (BT) toxin in susceptible strains. Also, Chang et al. (2017) knocked out a pheromone receptor, which validated its significant function in mating time regulation19. These reports suggest that CRISPR/Cas9 can act as an effective tool to study gene function in vivo in insect systems. However, a detailed procedure for CRISPR/Cas9 modification in insect systems remains incomplete, which limits its application range in insect functional genomics.
Here, we present a protocol for knocking out a functional gene in H. armigera using the CRISPR/Cas9 system. A detailed step-by-step protocol is provided, including the design and preparation of gene-specific primers for gRNA production, embryo collection, microinjection, insect rearing, and mutant identification. This protocol serves as a valuable reference to manipulate any functional genes in H. armigera and can be extended to other Lepidoptera species.
Protocol
1. Design of gene-specific primers and preparation of sgRNA
- Verify a conserved genomic region in the gene of interest through PCR amplification and sequencing analyses. Amplify the target gene from the genome DNA of H. armigera and distinguish the exons and introns.
NOTE: The sequence specificity of the guide site is necessary to avoid off-target gene editing. Search possible guide sites in the exons are close to the 5' UTR of the gene. Then, it is important to make sure that the gene is completely nonfunctional. A summary of the flow path for the preparation of sgRNA is illustrated in Figure 1. - Choose the sgRNA targets. Use the CRISPR online website CRISPOR (Version 4.97) (http://crispor.tefor.net/crispor.py) to search for possible guide sites in the exons close to the 5' UTR of the gene. Input the exon sequence into the textbox and select the target genome to Helicoverpa armigera (Harm_1.0). Choose the protospacer adjacent motif (PAM) option of "20 bp-NGG" and leave the other settings on the default parameters according to the user manuals of the websites.
- Compare the predicted guide sequences from the software and choose the guide sequence with the highest predicted efficiency and fewest mismatches to improve the editing efficiency and reduce off-target editing. A 20 bp guide sequence containing one or two G on the 5' UTR is recommended as it could increase the cutting efficiency.
NOTE: A pair of gRNAs across exon-regions are also recommended to obtain a large segment deletion, which simplifies mutant detection in later steps. Ensure the spacing distance between the two selected guide sequences is at least 100 bp. In this protocol, we choose the commonly used SpCas9 protein, which recognizes the NGG motif. According to the manufacturer's instruction, the guide sequence lacking G is also acceptable when choosing the T7 promoter because the promoter adds a G to the 5' UTR of the sequence. - Design forward and reverse oligonucleotides. Set the sequence order to 5'-20 bp guide sequence-NGG-3' and reverse complement the guide sequence. Add the T7 promoter sequence to the forward and reverse strand guide sequence, respectively according to the user guide of the gRNA synthesis kit.
NOTE: The PAM sequence NGG should be excluded from the oligonucleotide sequence. - Generate the sgRNA using the gRNA synthesis kit. This process includes three steps: DNA template assembly, in vitro transcription, and purification of sgRNA (Figure 2). Perform each step in accordance with the user instructions.
2. Embryo preparation and collection
- Separate male and female pupae as described by Hongtao et al.34 and segregate them into two different net cages. After eclosion, feed them ~30 mL of 10% (w/v) white sugar solution in absorbent cotton in a Petri dish.
NOTE: 3 g of white sugar was dissolved in 30 mL of sterile water to prepare the 10% (w/v) white sugar solution. - Select 50 healthy individuals from 3-day-old males and 2-day-old females, respectively, and mix them in a clean net cage. Place a piece of cotton containing 10% (w/v) sugar solution in the cage and keep the cotton moist. Cover the net cages with gauze and fix the gauze with a rubber band. Spray water onto the gauze to keep moist.
- Allow selected male and female moths in step 2 to mate completely and observe the egg-laying amount.
NOTE: The peak of oviposition of H. armigera appears after 9:00 p.m. Therefore, the time of the mating should be considered to ensure a sufficient number of eggs in the subsequent steps. At the peak of oviposition, replace the gauze with a black cloth and enable free oviposition for 30 min. Replace the black cloth every 30 min for collecting fresh eggs (Figure 3A). - Cut the black cloth into irregularly shaped patches with a size of 3 mm approximately. Ensure more eggs on each patch.
NOTE: Avoid choosing wrinkled eggs as they are usually unfertilized. - Paste double-sided tape onto a microscope slide (25 mm x 75 mm) (Figure 3B). Using forceps, paste the patches with eggs in a row on the surface of the double-sided tape. Press the margin of each patch to make sure they stick firmly to the tape. Collect 50-100 eggs per microscope slide (Figure 3C).
NOTE: The patches need to cover the full surface of the double-sided tape, otherwise the hatching larval will have difficulty crawling out. - Before microinjection, keep the microscope slide on ice to delay the development of embryos.
3. Microinjection of embryos
- Prepare the needle by pulling a capillary glass using a micropipette puller (Figure 4A). Set Heat to 540, Pull to 80, Vel to 75, Time to 170, and Pressure to 450. Ground the needle tip using a micro grinder. The ideal needle shows a sharp-edged tip (Figure 4B).
- Preparation of injection solutions. Add 2 µL of commercialized Cas9 protein (1 mg/mL) and sgRNA (300-500 ng/µL final concentration) to RNase-free water in a PCR tube to obtain a 10 µL volume mixture. The volume of sgRNA depends on its concentration. Mix well by pipetting and put it on ice.
NOTE: All the pipette tips and PCR tubes used in this step are RNase-free. - Set the parameters of the electronic microinjector. Set the injection pressure (pi) to 1,500 hPa, the injection time (ti) to 0.1 s, and the compensation pressure (pc) to 30 hPa.
- Load 2 µL of the mixture into a needle using a micro loader pipette tip. The residual air in the tip of the needle should be exhausted as much as possible.
- Connect the injection needle to a micromanipulator and ensure a tight connection between the two parts.
- Place a slide in a Petri dish (100 mm) and put them on the stage of the microscope (Figure 5A).
- Adjust the position of the needle tip under a light microscope until both the needle tip and embryos are visible under the microscope (Figure 5B).
- Adjust the volume of the droplet. Press the pedal and observe the liquid drop at the needle tip. Adjust the injection pressure of the microinjector until the volume of a liquid drop is about one-tenth of the volume of the embryos.
NOTE: The quality of the injection needle is vital for the survival rate of embryos. - Carefully insert the needle tip into the top hemisphere of an embryo at a 45-degree angle (Figure 5C). Press the pedal to deliver the mixture into the embryo. The injection leads to a slight expansion of the embryo. Retract the needle immediately from the embryo and move the Petri dish with one hand until the next embryo is in proximity to the needle and inject the next embryo with the same procedure.
NOTE: The cytoplasmic outflow at the pinhole is acceptable. If the cytoplasmic leakage is too much, adjust the angle of the needle to a more severe angle until the fluid outflow is controlled. - Inject at least 300 embryos to ensure a sufficient hatching amount. Cover the lid of the Petri dish(es) after injection.
NOTE: The time from oviposition to injection of the 50-100 embryos is limited to 2 h. Most of the embryos are still at the one-cell stage within this time frame. In general, it is useful to repeat the oviposition procedure during embryo injection to promote efficiency.
4. Post-injection insect rearing
- Propagation of G0 embryos.
- Incubate the embryos at 60% relative humidity and 28°C in an artificial climate box with 14 h light/10 h dark.
- Check the development of embryos daily after injection. When the surface color of embryos has darkened, put artificial diet in the Petri dish around the microscope slide, and check the development of embryos every 12 h.
NOTE: The artificial diet is prepared as described by Wu et al.35 and Jha et al.36. - Prepare 24-well culture plates and fill each well to one-third of its volumetric capacity with artificial diet.
- Pick out hatching larvae (Figure 5D) using a small paintbrush and transfer them to the 24-well culture plate. Insert one larva per well to ensure that each larva has enough food to survive.
NOTE: The larvae of H. armigera were reared individually in each well since they usually cannibalized each other.
- G0 larvae rearing
- Rear the larvae in the same conditions as the embryos.
- Check the hatched larvae every day. When larvae grow to the third instar stage, transfer each larva into a new glass dactylethrae, filling one-fifth of the volume with artificial diet.
- Approximately 12 d after incubation, the mature larvae should begin to pupate.
- The G0 mature pupae are distinguished based on sex and placed in separate cages before eclosion. The same-aged pupae of wild type are also prepared.
- G0 adults rearing
- Check the eclosion of both wild type and mutant pupae daily.
- Transfer the newly-eclosed G0 male adults and wild type female adults into a fresh net cage and make the ratio between G0 and wild type at approximately 1:1. Supply them with 10% (w/v) sugar solution dropped in cotton balls.
- Rear insects using the routine method above until the pupation of generation one (G1).
- Using a dactylethrae, transfer the newly-eclosed single pair of G1 adults into a plastic jar (13 cm x 12 cm x 12 cm) supplied with 10% (w/v) sugar solution. Cover each jar with gauze. Take about 50 pairs of G1 adults in total.
NOTE: Eclosion of female moths predate that of male moths. In general, a 3-day-old male and a 2-day-old female are sexually mature and ready to mate. The newly-eclosed adults will not mate in their first light period without feeding. - Collect the G1 adults in a plastic jar after they have laid eggs. Put each adult moth in a 1.5 mL centrifuge tube.
5. Knock-out mutant detection
- Design a pair of primers spanning the predicted truncated site. The primers should be set at least 50 bp distance on either side (upstream and downstream) from the target site.
NOTE: The primers for the identification of the target sequence often cover large spans to amplify efficiently. - Perform the PCR reaction using the genotyping primers with genome DNA extracted in section 1. Perform PCR cycling at 95 °C for 20 s; 35 cycles of 95 °C for 20 s, 55 °C for 20 s, 72 °C for 1 min; 72 °C for 5 min, and 4 °C on hold. Verify the reaction product via 1% (w/v) agarose gel. The selected pair of detection primers was confirmed by the quality of the PCR product. If the bands are evident and specific, the primers could be used for further mutant detection based on amplicon size.
- Screen for potential edited individuals. Remove a hind leg carefully using forceps and put each leg in a lysing matrix tube, respectively. Label the lysing matrix tube consistent with the number on the glass dactylethrae.
- Homogenize the hind leg using a tissue homogenizer. Set the speed to 6.0 m/s and the time to 60 s. Extract the genomic DNA of the homogenized sample using a commercial gDNA extraction kit according to the manufacturer's instructions.
- Amplify the gene segment using genotyping primers with the same PCR reaction conditions as described in step 2. Confirm the genotype by a gene sequencing service. Once G1 mutant genotypes (target the same site) contained in the same jar were detected, keep the G1 progeny and rename it as generation two (G2).
- Put G1 individuals of the same genotype in one net cage. Self-cross the G1 progenies and continue to screen using the same methods.
- Amplify the gene segment and confirm the genotype with the same procedure as outlined in step 2. Obtain G2 homozygous lines and maintain the knock-out mutant line.
Representative Results
This protocol provides detailed steps for obtaining gene knock-out lines of H. armigera using CRISPR/Cas9 technology. The representative results obtained by this protocol are summarized for gDNA selection, embryo collection and injection, insect rearing, and mutant detection.
In this study, the target site of our gene of interest was located in its second exon (Figure 2A). This site was highly conserved, and the target band fragment of synthesized sgRNA was confirmed using agarose gel electrophoresis (Figure 2B,C,D).
The male and female moths were initially reared in separate net cages to prevent mating ahead of schedule and to ensure a sufficient quantity of embryos as much as possible. In general, a total number of 300 fertilized eggs were collected and were immediately injected with the sgRNA/Cas9 protein mixture (300-500 ng/µL of sgRNA, 200 ng/µL of Cas9 protein) at the one-cell stage. The injection volume was about one-tenth that of the embryos. After microinjection, the embryos were reared as described in section 4, and 40%-60% of injected embryos survived.
The mutant detection of a single sgRNA target was performed by sequencing the PCR products from G1 parental adults (Figure 6B). We also tested the effectiveness of using non-overlapping sgRNA pairs across different exons. The large deletion of the mutants (Figure 6C,D) can be easily distinguished from wild type bands (Figure 6A).
The mutation rate calculated in this protocol was 87.50% when 16 individuals are randomly tested, indicating that this protocol was highly-efficient. Gene knockout results were shown in several genotypes, but the majority of mutants identified from our screening were -2 bp type. Mutations resulted in the premature termination of protein translation in the genome, which subsequently led to the loss of gene function.
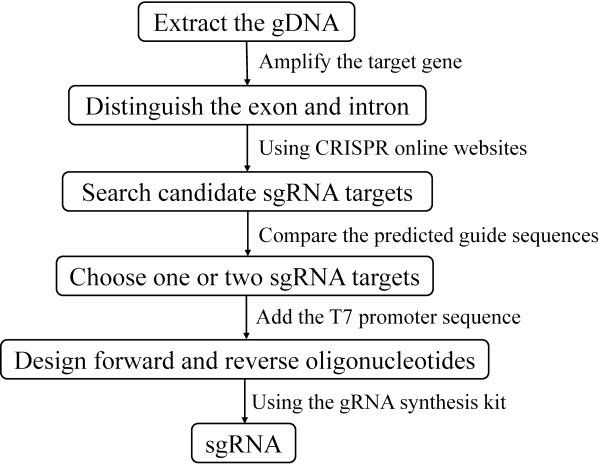
Figure 1: The flowchart for the preparation of sgRNA. Please click here to view a larger version of this figure.
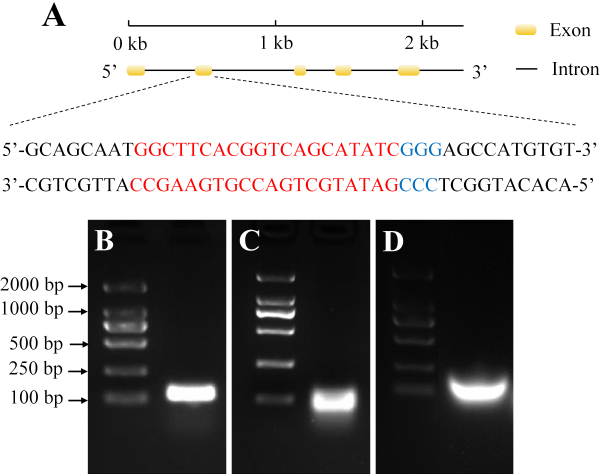
Figure 2: Selection and synthesis of target sgRNAs from H. armigera. (A) The yellow domain represents the exon, while the black line represents the intron. The red sequences indicate the target sequence, and the blue sequences indicate the protospacer adjacent motif (PAM). (B) PCR assembly of the sgRNA DNA template. (C) The in vitro transcription product. (D) Purification of sgRNA. Please click here to view a larger version of this figure.

Figure 3: Embryo collection. (A) A net cage covered with black cloth. The male and female moths of H. armigera were mating. (B) The microscope slide without embryos. (C) The microscope slide containing 50-100 embryos on pieces of black cloth. Please click here to view a larger version of this figure.

Figure 4: Needle preparation. (A) Micropipette puller. (B) Tip of a microinjection needle after pulling by a micropipette puller. The dotted box indicates the magnified needle tip. Scale bar represents 1 mm. Please click here to view a larger version of this figure.

Figure 5: Embryo microinjections. (A) The whole set of a microinjection system containing a microscope (middle) and an electronic microinjector (left) connected to a micromanipulator (right). (B) Embryos and microinjection needle. (C) The injection site of the embryo is labeled with the red arrow. Scale bar represents 200 µm. (D) A hatched larva under the microscope. Scale bar represents 1 mm. Please click here to view a larger version of this figure.
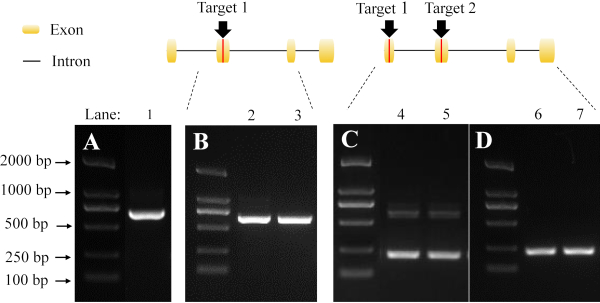
Figure 6: Detection of mutants by PCR and gel electrophoresis. The black arrows and red lines indicate the target sites of the sgRNA. (A) The band in lane 1 represents the amplification fragment derived from wild type. (B) The bands in lane 2 and 3 represent the amplification fragment derived from mutant using a single sgRNA target. (C) The detection of a heterozygote using a pair of non-overlapping sgRNA. The bands in lanes 4 and 5 represent the amplification fragment derived from the mutation of two sgRNA targets. The lower bands indicate a large fragment deletion. (D) The results are derived from a homozygote. The bands in lane 6 and 7 indicate the large fragment deletion. Please click here to view a larger version of this figure.
Discussion
The application of the CRISPR/Cas9 system has provided powerful technical support for the analysis of gene function and interaction among various genes. The detailed protocol we present here demonstrates the generation of a homozygote mutant in H. armigera via CRISPR/Cas9 genome editing. This reliable procedure provides a straightforward way for directed gene mutagenesis in H. armigera.
The choice of CRISPR target sites could affect the mutagenesis efficiency37. In this protocol, we compared and analyzed multiple results from the online website CRISPOR to obtain an appropriate target site. In silico, gRNA predictions present some advantages. Firstly, they analyze the whole genome of H. armigera when designing sgRNAs to minimize the off-target effects. The online resources mentioned above, as well as CHOPCHOP (http://chopchop.cbu.uib.no/), function with a number of Lepidoptera genomes, which could be beneficial for gene editing in other Lepidopteran moths. Secondly, the ranking of the candidate sgRNAs directly compares possibilities but might include some variations based on the different algorithms. The candidate sequence with high ratings in both lists tends to be more reliable. However, a major limitation of this protocol is that a large number of insect genomes are absent in the databases of the websites, so there is potential for off-target effects. Another limitation is that the PAM sequence is necessary for the sgRNA design, which may result in the inability to find an appropriate target site.
The tissues used for mutant screening are also a crucial factor. The survival rate, life cycle, and physiological functions of insects should not be affected. In our process of exploring the optimal gDNA extraction method, the micro-hemolymph extraction from larvae was attempted for mutant detection to save time and avoid mating (unpublished data). However, this method brought more challenges regarding the efficiency of PCR amplification and the survival rates of adult (data not shown). In addition, Zheng et al.38 reported a non-destructive method for gDNA extraction using the exuviate or puparia. Based on those findings, we modified and explored an approach using hind legs for gDNA extraction, which allows adult moths to survive and mate naturally, significantly improving the detection accuracy of a given genotype. Therefore, we developed a new method to increase the success rate of gDNA extraction by removing one of the hind legs from each adult candidate. We further confirmed that this operation did not affect the survival rate and mating frequency of adult moths. Furthermore, we found that the large fragment deletion can be easily observed by the gel electrophoresis when co-injected with a pair of gRNAs across exon-regions (Figure 6), which simplifies the process of mutant identification when screening.
The eggs of H. armigera are collected on a black cloth, which makes it easy to distinguish the eggs under the microscope in the process of microinjection (Figure 5B). Due to the common reproductive behaviors of Lepidopteran moths such as mating, oviposition, hatching, and eclosion39,40,41, this egg-collecting technique could also be applied for other Lepidopteran moths.
In conclusion, the CRISPR/Cas9 system has proven to be a reliable tool for facilitating functional genomics studies in H. armigera. The step-by-step descriptions enable users to complete an integral gene-editing process.
Declarações
The authors have nothing to disclose.
Acknowledgements
This work was supported by National Natural Science Foundation of China (31725023, 31861133019 to GW, and 31171912 to CY).
Materials
| 2kb DNA ladder | TransGen Biotech | BM101 | |
| Capillary Glass | World Precision Instrucments | 504949 | referred to as "capillary glass" in the protocol |
| Double Sided Tape | Minnesota Mining and Manufacturing Corporation | 665 | |
| Eppendorf FemtoJet 4i Microinjector | Eppendorf Corporate | E5252000021 | |
| Eppendorf InjectMan 4 micromanipulator | Eppendorf Corporate | 5192000051 | |
| Eppendorf Microloader Pipette Tips | Eppendorf Corporate | G2835241 | |
| GeneArt Precision gRNA Synthesis Kit | Thermo Fisher Scientific | A29377 | |
| Microscope Slide | Sail Brand | 7105 | |
| Olympus Microscope | Olympus Corporation | SZX16 | |
| PrimeSTAR HS (Premix) | Takara Biomedical Technology | R040 | used for mutant detection |
| Sutter Micropipette Puller | Sutter Instrument Company | P-1000 | |
| TIANamp Genomic DNA Kit | TIANGEN Corporate | DP304-03 | |
| TrueCut Cas9 Protein v2 | Thermo Fisher Scientific | A36499 |
Referências
- Cong, L., et al. Multiplex genome engineering using CRISPR/Cas systems. Science. 339 (6121), 819-823 (2013).
- Garneau, J. E., et al. The CRISPR/Cas bacterial immune system cleaves bacteriophage and plasmid DNA. Nature. 468 (7320), 67-71 (2010).
- Jinek, M., et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science. 337 (6096), 816-821 (2012).
- Doudna, J. A., Charpentier, E. The new frontier of genome engineering with CRISPR-Cas9. Science. 346 (6213), 1258096 (2014).
- Ding, Q., et al. Enhanced efficiency of human pluripotent stem cell genome editing through replacing TALENs with CRISPRs. Cell Stem Cell. 12 (4), 393-394 (2013).
- Collins, P. J., Hale, C. M., Xu, H. Edited course of biomedical research: leaping forward with CRISPR. Pharmacological Research. 125, 258-265 (2017).
- Huang, J., Wang, Y., Zhao, J. CRISPR editing in biological and biomedical investigation. Journal of Cellular Physiology. 233 (5), 3875-3891 (2018).
- Guan, L., Han, Y., Zhu, S., Lin, J. Application of CRISPR-Cas system in gene therapy: pre-clinical progress in animal model. DNA Repair. 46, 1-8 (2016).
- Wu, J., et al. Gene therapy for glaucoma by ciliary body aquaporin 1 disruption using CRISPR-Cas9. Molecular Therapy. 28 (3), 820-829 (2020).
- Jiao, R., Gao, C. The CRISPR/Cas9 genome editing revolution. Journal of Genetics and Genomics. 43, 227-228 (2016).
- Gupta, M., Gerard, M., Padmaja, S. S., Sastry, R. K. Trends of CRISPR technology development and deployment into Agricultural Production-Consumption Systems. World Patent Information. 60, 101944 (2020).
- Es, I., et al. The application of the CRISPR-Cas9 genome editing machinery in food and agricultural science: Current status, future perspectives, and associated challenges. Biotechnology Advances. 37 (3), 410-421 (2019).
- Tarasava, K., Oh, E. J., Eckert, C. A., Gill, R. T. CRISPR-enabled tools for engineering microbial genomes and phenotypes. Biotechnology Journal. 13 (9), 1700586 (2018).
- Ma, X., Zhu, Q., Chen, Y., Liu, Y. -. G. CRISPR/Cas9 platforms for genome editing in plants: developments and applications. Molecular Plant. 9 (7), 961-974 (2016).
- Pandey, P. K., et al. Versatile and multifaceted CRISPR/Cas gene editing tool for plant research. Seminars in Cell & Developmental Biology. 96, 107-114 (2019).
- Friedland, A. E., et al. Heritable genome editing in C. elegans via a CRISPR-Cas9 system. Nature Methods. 10 (8), 741-743 (2013).
- Mojica, F. J., Montoliu, L. On the origin of CRISPR-Cas technology: from prokaryotes to mammals. Trends in Microbiology. 24 (10), 811-820 (2016).
- Taning, C. N. T., Van Eynde, B., Yu, N., Ma, S., Smagghe, G. CRISPR/Cas9 in insects: Applications, best practices and biosafety concerns. Journal of Insect Physiology. 98, 245-257 (2017).
- Chang, H., et al. A pheromone antagonist regulates optimal mating time in the moth Helicoverpa armigera. Current Biology. 27 (11), 1610-1615 (2017).
- Yan, H., et al. An engineered orco mutation produces aberrant social behavior and defective neural development in ants. Cell. 170 (4), 736-747 (2017).
- Koutroumpa, F., et al. Heritable genome editing with CRISPR/Cas9 induces anosmia in a crop pest moth. Scientific Reports. 6 (1), 29620 (2016).
- Chen, D., et al. CRISPR/Cas9-mediated genome editing induces exon skipping by complete or stochastic altering splicing in the migratory locust. BMC Biotechnology. 18 (1), 1-9 (2018).
- Fitt, G. P. The ecology of Heliothis species in relation to agroecosystems. Annual Review of Entomology. 34 (1), 17-53 (1989).
- Jallow, M. F., Cunningham, J. P., Zalucki, M. P. Intra-specific variation for host plant use in Helicoverpa armigera (Hübner)(Lepidoptera: Noctuidae): implications for management. Crop Protection. 23 (10), 955-964 (2004).
- Ai, D., et al. Gene cloning, prokaryotic expression, and biochemical characterization of a soluble trehalase in Helicoverpa armigera Hübner (Lepidoptera: Noctuidae). Journal of Insect Science. 18 (3), 22 (2018).
- Wan, F., et al. A chromosome-level genome assembly of Cydia pomonella provides insights into chemical ecology and insecticide resistance. Nature Communications. 10 (1), 1-14 (2019).
- Cheng, T., et al. Genomic adaptation to polyphagy and insecticides in a major East Asian noctuid pest. Nature Ecology & Evolution. 1 (11), 1747-1756 (2017).
- Xu, W., Papanicolaou, A., Zhang, H. J., Anderson, A. Expansion of a bitter taste receptor family in a polyphagous insect herbivore. Science Reports. 6, 23666 (2016).
- Gouin, A., et al. Two genomes of highly polyphagous lepidopteran pests (Spodoptera frugiperda, Noctuidae) with different host-plant ranges. Scientific Reports. 7 (1), 1-12 (2017).
- Wang, J., et al. Functional validation of cadherin as a receptor of Bt toxin Cry1Ac in Helicoverpa armigera utilizing the CRISPR/Cas9 system. Insect Biochemistry and Molecular Biology. 76, 11-17 (2016).
- Wang, J., Wang, H., Liu, S., Liu, L., Wu, Y. CRISPR/Cas9 mediated genome editing of Helicoverpa armigera with mutations of an ABC transporter gene HaABCA2 confers resistance to Bacillus thuringiensis Cry2A toxins. Insect Biochemistry and Molecular biology. 87, 147 (2017).
- Wang, J., Ma, H., Zhao, S., Huang, J., Wu, Y. Functional redundancy of two ABC transporter proteins in mediating toxicity of Bacillus thuringiensis to cotton bollworm. PLoS Pathogens. 16 (3), 1008427 (2020).
- Jin, L., et al. Dominant point mutation in a tetraspanin gene associated with field-evolved resistance of cotton bollworm to transgenic Bt cotton. Proceedings of the National Academy of Sciences. 115 (46), 11760-11765 (2018).
- Hongtao, Z., Jianan, L., Lian, T., Yang, Z. Sexing of Helicoverpa assulta and Helicoverpa armigera pupae based on machine vision. Tobacco Sciene & Technology. 53 (2), 21-26 (2019).
- Wu, K., Gong, P. A new and practical artificial diet for the cotton boll-worm. Insect science. 4 (3), 277-282 (1997).
- Jha, R. K., Chi, H., Li-Cheng, T. A Comparison of Artificial Diet and Hybrid Sweet Corn for the Rearing of Helicoverpa armigera (Hübner) (Lepidoptera: Noctuidae) Based on Life Table Characteristics. Environmental Entomology. (1), 30 (2012).
- Bassett, A. R., Tibbit, C., Ponting, C. P., Liu, J. -. L. Highly efficient targeted mutagenesis of Drosophila with the CRISPR/Cas9 system. Cell Reports. 4 (1), 220-228 (2013).
- Zheng, M. -. Y., et al. A non-destructive method of genotyping individual insects from the exuviate of final instar larvae, or puparia. Chinese Journal of Applied Entomology. (2), 21 (2018).
- Pashley, D. P., Hammond, A. M., Hardy, T. N. Reproductive isolating mechanisms in fall armyworm host strains (Lepidoptera: Noctuidae). Annals of The Entomological Society of America. 85 (4), 400-405 (1992).
- Kamimura, M., Tatsuki, S. Diel rhythms of calling behavior and pheromone production of oriental tobacco budworm moth, Helicoverpa assulta(Lepidoptera: Noctuidae). Journal of Chemical Ecology. 19 (12), 2953-2963 (1993).
- Edwards, K. D. Activity rhythms of lepidopterous defoliators: ii. Halisidota argentata pack. (arctiidae), and nepytia phantasmaria stkr. (GEOMETRIDAE). Canadian Journal of Zoology. 42 (6), 939-958 (1964).

