Stimulating Nerve Stem Cell Differentiation Using Cold Atmospheric Plasma Treatment
Abstract
Source: Xiong, Z., et al. Nerve stem cell differentiation by a one-step cold atmospheric plasma treatment in vitro. J. Vis. Exp. (2019).
This video presents a method for triggering differentiation in neural stem cells by utilizing cold atmospheric plasma. This generates UV radiation and reactive molecules to stimulate nerve cells to produce projections, indicating neuronal cell differentiation.
Protocol
1. Cell Cultures and Predifferentiation
- Neural stem cell culture and predifferentiation
- Prepare poly-D-lysine-coated coverslips. Put a sterile coverslip (20 mm in diameter) into a 12-well plate. Coat the cover glass with poly-D-lysine, 0.1% w/v, in water for better cell adhesion on the coverslips by following the next steps.
NOTE: Optimal conditions must be determined for each cell line and application.- Aseptically coat the surface of the coverslip with poly-D-lysine, 0.1% w/v, in water. Rock gently to ensure an even coating of the coverslip surface.
- After culturing overnight (~12 h) at 37°C, remove the poly-D-lysine solution and rinse the surface 3x with 1 mL of sterile water.
- Dry the cells at least 30 min before seeding them.
- Prepare poly-D-lysine-coated coverslips. Put a sterile coverslip (20 mm in diameter) into a 12-well plate. Coat the cover glass with poly-D-lysine, 0.1% w/v, in water for better cell adhesion on the coverslips by following the next steps.
- Murine neural stem cell C17.2 (C17.2-NSC) culture and predifferentiation
- Incubate C17.2-NSCs in a 25 cm2 flask in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum (FBS), 5% horse serum (HS), and 1% penicillin/streptomycin at 37 °C and 5% CO2 for ~2 – 3d.
- When the cells reach 85% confluence, remove the medium, wash the cells with 1 mL of PBS, and add 1 mL of fresh trypsin (0.25%) into the flask. Leave it for 1 min; then, add 1 mL of culture medium to the flask and pipette up and down several times to ensure a single-cell suspension.
- Count the density of the cells in the suspension using a hemocytometer. Calculate the required volume of cell suspension to give a final cell concentration of 2 x 104 cells/mL.
- Seed the C17.2-NSCs on the coated coverslips in the 12-well plate with a density of ~2 x 104 cells per well. Check the cell density under a microscope.
NOTE: For a better result, the optimal cell density must be determined before the immunofluorescence experiment. - Incubate the cells at 37°C in a cell incubator for 12 h to allow for attachment.
- After attachment, wash the cells 2x with 1 mL of PBS and cultivate the cells with a differentiation medium consisting of DMEM/F12 with 1% N2 supplement for 48 h before the plasma treatment.
- Primary rat neural stem cell culture and predifferentiation
- Culture the primary rat NSC (neural stem cells) suspension in rat NSC growth medium in uncoated T25 flasks at a density of 5 x 105 cells.
- Check the formation of neurospheres under an inverted microscope. Make sure the morphology of the neurospheres exhibits spherical and transparent multicellular complexes.
- When the neurospheres reach a diameter of 3 mm or larger, transfer the neurosphere suspension to a 15 mL sterile centrifuge tube and let the neurospheres settle by gravity.
- Remove the supernatant carefully to leave the neurospheres in a minimal volume of medium.
- Rinse the neurospheres 1x with 5 mL of PBS and let the neurospheres settle by gravity. Remove the supernatant to leave a minimal volume of PBS.
- Add a suitable volume of differentiation medium to adjust the cell density to ~12 – 15 neurospheres per milliliter. The differentiation medium for primary rat NSCs consists of DMEM/F12, 2% B27, and 1% FBS.
- Seed 1 mL of neurosphere suspension onto each coated coverslip in the 12-well plate.
NOTE: Shake the plate gently to ensure that the neurospheres are evenly distributed. This step is critical for a better immunofluorescence result. - Place the plate into the incubator and allow it to predifferentiate for 24 h before the plasma treatment.
2. Preparation of the Plasma Jets
- Choose a half-open quartz tube with an internal diameter of 2 mm and an external diameter of 4 mm. Insert a high-voltage wire with a diameter of 2 mm into the tube.
- Insert the quartz tube with the high-voltage wire into a 5 mL syringe and use a holder to mount it inside the center of the syringe. Set the distance between the sealed end of the quartz tube and the syringe tip at 1 cm.
- Connect a 1 m silicone rubber pipe (with an inner diameter of 12 mm) to the open end of the syringe and then, connect it to the flowmeter and gas valve in sequence.
3. Acquisition of the Jets
- Connect the circuit as shown in Figure 1. Connect the output wire of the power supply to the plasma jet device and then, connect the tip of the high-voltage probe with the output wire to detect the voltage. Connect the other end of the high-voltage probe to the oscilloscope to record the information of the output voltage. Check the whole circuit and make sure the power supply, the oscilloscope, and the high-voltage probe are all grounded.
- Check the gas line. Make sure the gas tube has been connected to the plasma jet device; then, open the gas valve of the helium (volume fraction, 99.999%) and oxygen (volume fraction, 99.999%) and set the gas flow to 1 L : 0.01 L/min (He:O2).
NOTE: Let the gas flow for several minutes before turning on the power supply for the first time. - Set the pulse amplitude, frequency, and pulse width as 8 kV, 8 kHz, and 1600 ns, respectively. Check the circuit again and then, turn on the output button to create a plasma jet.
CAUTION: Do not touch the high-voltage wire at any time.
4. Plasma Treatment of Neural Stem Cell
- Set the distance between the nozzle of the syringe and the cell well hole to 15 mm.
NOTE: The distance is measured from the bottom of the platform where the 12-well plate is placed. - Take the 12-well plate out of the incubator and change the medium of the predifferentiated cells to 800 µL of fresh differentiation medium.
- Divide the cells into three groups: an untreated control group, a 60 s He-and-O2 (1%) gas flow treatment group, and a 60 s plasma treatment group.
NOTE: There is no obvious liquid loss under the treatment condition. - Place the 12-well plate under the plasma jets, make sure the syringe nozzle is fixed in the center of each hole, and give the relevant treatment to the different groups mentioned in step 4.3.
NOTE: The untreated controls were kept in differentiation medium at room temperature during the experimental procedure to ensure uniform treatment conditions. The He-and-O2 (1%) gas flow treatment group was treated with only a He and O2 (1%) gas flow, without plasma generation. All the treatments should be performed in triplicate.
5. Neural Stem Cell Differentiation
- After treatment, remove the original culture and add 1 mL of new differentiation medium to each well.
- Incubate the cells in the incubator at 37°C and 5% CO2 for 6 d. Change the medium every other day.
- Check the differentiation status of the different groups daily under an inverted phase-contrast light microscope Figure 2. Randomly select at least 12 fields and take photos to record the morphological changes.
Representative Results
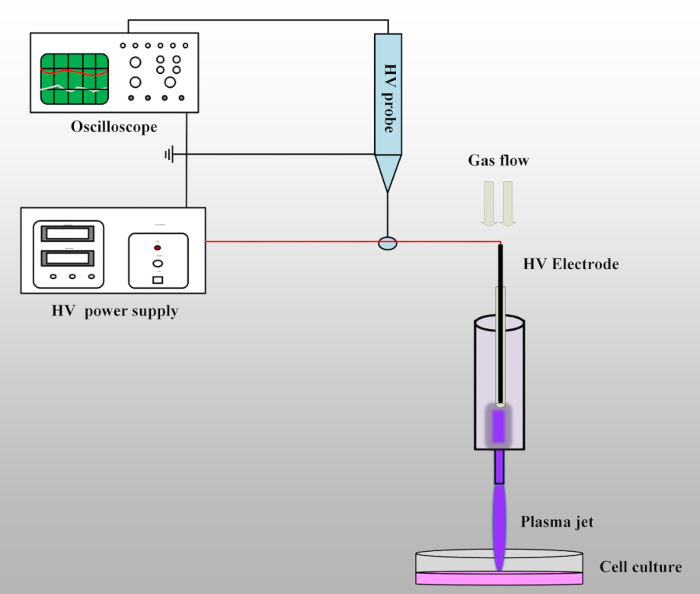
Figure 1: Schematic of the experimental set-up. The plasma jet device is connected to the output wire of the high-voltage power supply. The high-voltage probe with oscilloscope is used to detect the output voltage. When the working gases are flowing through the syringe and the high voltage is on, the plasma jet will be generated and propagate into the open air.
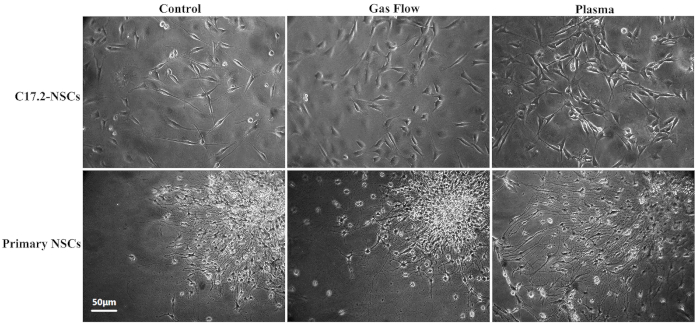
Figure 2: Ordinary inverted phase-contrast light microscope images for C17.2-NSCs and primary rat NSCs. This figure is adapted from Xiong et al. with permission.
Divulgaciones
The authors have nothing to disclose.
Materials
| Coverslip | NEST | 801008 | |
| Poly-D-lysine | Beyotime | P0128 | |
| DMEM medium | HyClone | SH30022.01B | stored at 4 °C |
| DMEM/F12 medium | HyClone | SH30023.01B | stored at 4 °C |
| N2 supplement | Gibco | 17502048 | stored at -20 °C and protect from light |
| B27 supplement | Gibco | 17504044 | stored at -20 °C and protect from light |
| Fetal bovine serum | HyClone | SH30084.03 | stored at -20 °C, avoid repeated freezing and thawing |
| Donor Horse serum | HyClone | SH30074.03 | tored at -20 °C, avoid repeated freezing and thawing |
| Penicillin/Streptomycin | HyClone | SV30010 | stored at 4 °C |
| Trypsin | HyClone | 25300054 | stored at 4 °C |
| PBS solution | HyClone | SH30256.01B | stored at 4 °C |
| 4% paraformaldehyde | Beyotime | P0098 | stored at -20 °C |
| TritonX-100 | Sigma | T8787 | |
| Light microscope | Nanjing Jiangnan Novel Optics Company | XD-202 | |
| High – voltage Power Amplifier | Directed Energy | PVX-4110 | |
| DC power supply | Spellman | SL1200 | |
| Function Generator | Aligent | 33521A | |
| Oscilloscope | Tektronix | DPO3034 | |
| High voltage probe | Tektronix | P6015A | |
| 12-well plate | corning | 3512 | |
| 25 cm2 flask | corning | 430639 | |
| Helium gas | |||
| Oxygen Gas | |||
| Plasma jet nozzel | |||
| CO2 Incubator | |||
| Silicone pipe | inner diameter of 12 mm | ||
| Murine neural stem cell C17.2 (C17.2-NSC) |

