에반스 방법
English
COMPARTILHAR
Visão Geral
출처: 타마라 M. 파워스, 텍사스 A&M 대학교 화학학과
대부분의 유기 분자는 diamagnetic, 모든 전자는 결합에 결합 하는 경우, 많은 전이 금속 복합체는 파라마그네틱, 결합 되지 않은 전자와 지상 상태를 가지고. 비슷한 에너지의 궤도의 경우 전자가 페어링하기 전에 페어링되지 않은 전자의 수를 최대화하기 위해 궤도를 채울 것이라고 명시한 Hund의 규칙을 기억하십시오. 전이 금속은 부분적으로 d-궤도를채우고 있으며, 에너지는 리간드를 금속에 조정하여 다양한 범위로 분반됩니다. 따라서, d-orbitals는 서로 에너지가 유사하지만 모두 퇴화되는 것은 아닙니다. 이를 통해 모든 전자가 페어링된 모든 전자 또는 파라마그네틱과 페어링되지 않은 전자와 함께 복합체가 다이자성으로 변할 수 있습니다.
금속 복합체에서 페어링되지 않은 전자의 수를 알면 금속 복합체의 산화 상태 및 기하학뿐만 아니라 리간드의 리간드 필드(crystal field) 강도에 대한 단서를 제공할 수 있습니다. 이러한 특성은 전이 금속 복합체의 분광학 및 반응성에 큰 영향을 미치므로 이해하는 것이 중요합니다.
페어링되지 않은 전자의 수를 계산하는 한 가지 방법은 조정 화합물의 자기 감수성, θ를측정하는 것입니다. 자기 감수성은 적용된 자기장에 배치할 때 물질(또는 화합물)의 자화의 척도이다. 쌍전자는 적용된 자기장에 의해 약간 격퇴되고, 자기장의 강도가 증가함에 따라 이 반발은 선형적으로 증가합니다. 한편, 페어링되지 않은 전자는 자기장에 (더 큰 범위까지) 끌리며, 매력은 자기장 강도로 선형적으로 증가합니다. 따라서, 결합되지 않은 전자를 가진 모든 화합물은 자기장에 끌릴 것입니다. 1
자기 감수성을 측정할 때, 우리는 자기 순간으로부터 짝을 이루지 않은 전자의 수에 대한 정보를 μ. 자기 감수성은 수학식 12에의해 μ 자기 모멘트와 관련이있습니다.
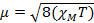 (1)
(1)
상수 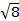 = [(3k B)/Nβ2],전자의 β= 보르 자그톤 (0.93 x 10-20 erg gauss-1),N = 아보가드로의 수 및 kB = 볼츠만 상수
= [(3k B)/Nβ2],전자의 β= 보르 자그톤 (0.93 x 10-20 erg gauss-1),N = 아보가드로의 수 및 kB = 볼츠만 상수
XM= 어금니 자기 감수성 (cm3/mol)
T = 온도(K)
μ = 자기 모멘트, 보르 자력 단위로 측정, μB = 9.27 x 10-24 JT-1
복합체의 자기 모멘트는 방정식 21에의해 주어집니다.
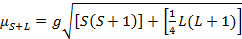 (2)
(2)
g = 자이로자성 비 = 2.00023 μB
S = 스핀 양자 번호 = ∑ms = [짝을 이루는 전자 수, n]/2
L = 궤도 양자 번호 = ∑ml
이 방정식에는 궤도 및 스핀 기여도가 모두 있습니다. 첫 번째 행 전환 금속 복합체의 경우 궤도 기여도가 작기 때문에 생략할 수 있으므로 회전 전용 자기 모멘트는 방정식 3에 의해 제공됩니다.
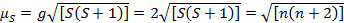 (3)
(3)
따라서 스핀 전용 자기 모멘트로 페어링되지 않은 전자의 수를 직접 제공할 수 있습니다. 이 근사치는 무거운 금속을 위해 이루어질 수 있지만, 궤도 기여는 두 번째 및 세 번째 열 전환 금속에 대한 중요 할 수 있습니다. 이 기여는 화합물이 그것 보다는 더 짝이 없는 전자가 있는 것처럼 보일 정도로 자기 순간을 팽창시키는 너무 중요할 지도 모릅니다. 따라서 이러한 복합체에 대해 추가 특성화가 필요할 수 있습니다.
본 실험에서, 트라이(acetylacetonato)의 용액 자기 모멘트(acetylacetonato)(III) (Fe(acac)3)은클로로폼에서 에반스 방법을 실험적으로 사용하여 결정된다.
Princípios
Procedimento
Resultados
Experimental Results
| Fe(acac)3 | Chloroform | |
| m (g) | 0.0051 | 0.874 |
| MW (g/mol) | 353.17 | n/a |
| n (mol) | 1.44⋅10–5 | n/a |
| Density (g/mL) | n/a | 1.49* |
| Volume (mL) | n/a | 0.587 |
| c (mol/mL) | 2.45⋅10-5 | |
| NMR shifts | Peak 1 | Peak 2 |
| δ (ppm) | 7.26 | 5.85 |
| Δppm | 1.41 | |
| NMR Instrument | ||
| Temperature (K) | 296.3 | |
| Field, F (Hz) | 500⋅106 |
* the density of the solvent can be approximated to the density of the solvent used
Calculations:

 = 0.0137 cm3/mol
= 0.0137 cm3/mol
 = 5.70 µB
= 5.70 µB
Theoretical Results for Given S and n Values:
| S | n | μS |
| 1/2 | 1 | 1.73 |
| 1 | 2 | 2.83 |
| 3/2 | 3 | 3.87 |
| 2 | 4 | 4.90 |
| 5/2 | 5 | 5.92 |
For 4.5 mg of Fe(acac)3 dissolved in 0.58 mL solvent, with a 300 MHz instrument a peak separation of 1.41 ppm is observed, which gives XM= 1.37 x 10-2 and µeff = 5.70. This µeff value is consistent with an S = 5/2 complex, which has 5 unpaired electrons.
Applications and Summary
The Evans method is a simple and practical method for obtaining the magnetic susceptibility of soluble metal complexes. This provides the number of unpaired electrons in a metal complex, which is pertinent to the spectroscopy, magnetic properties, and reactivity of the complex.
Measuring the magnetic susceptibility of paramagnetic species gives the number of unpaired electrons, which is a key property of metal complexes. As the reactivity of metal complexes is influenced by its electronic structure – that is, how the d-orbitals are populated – it is important to establish the number of unpaired electrons. The magnetic susceptibility can be used to determine the geometry of the metal complex in solution, give insight into the ligand field strength, and can provide evidence for the correct formal oxidation-state assignment of the metal complex. In the modules on "Group Theory" and "MO Theory of Transition Metal Complexes," we will introduce how to predict d-orbital splitting diagrams as well as how to use data from the Evans method to help determine the geometry of a metal complex and provide evidence for the oxidation state of the metal center.
There are multiple instruments that can be used to measure the magnetic susceptibility of a paramagnetic species including a Gouy balance, SQUID, or NMR instrument. The Evans method is a simple and practical technique that uses NMR to determine the solution magnetic moment of a paramagnet. While the Evans method is a powerful tool in the field of magnetism, there are several drawbacks to the technique. First, the molecule must be soluble in the solvent used in the experiment. If the paramagnetic sample is not fully dissolved, the concentration of the solution will be incorrect, which will lead to errors in the experimentally determined solution magnetic moment. Other errors in concentration can arise if the paramagnetic sample has diamagnetic (solvent) or paramagnetic impurities.
Referências
- Miessler, G. L., Fischer, P. J., Tarr, D. A. Inorganic Chemistry. 5 ed. Pearson. (2014).
- Drago, R. S. Physical Methods for Chemists. 2 ed. Saunders College Publishing. (1992).
- Girolami, G. S., Rauchfuss, T. B., Angelici, R. J. Synthesis and Technique in Inorganic Chemistry: A Laboratory Manual. 3 ed. University Science Books. Sausalito, CA, (1999).
Transcrição
The Evans method is a technique for calculating the number of unpaired electrons in solution-state metal complexes.
Many transition metal complexes have unpaired electrons, making them attracted to magnetic fields. These complexes are called paramagnetic. Complexes with all paired electrons are called diamagnetic.
Knowing the number of unpaired electrons is important for predicting the reactivity of a compound. The Evans method uses NMR spectroscopy to measure the parameters needed to calculate the number of unpaired electrons.
This video will illustrate the procedure for performing the Evans method, demonstrate the analysis of Fe(acac)3, and introduce a few applications of counting unpaired electrons in chemistry.
The number of unpaired electrons in a complex can be determined from the magnetic moment of the given molecule. The magnetic moments of 1st row transition metal complexes can be approximated from the contributions of unpaired electrons, called the spin-only magnetic moment. For the 2nd and 3rd row transition metal complexes, both the spin and orbital contributions must be considered.
The magnetic moment is related to the magnetic susceptibility, which provides the degree of magnetization of a complex in an applied magnetic field.
The chemical shift of a species in an NMR spectrum is affected by the overall magnetic susceptibility of the sample solution. Thus, the chemical shift of a solvent changes if the solute is paramagnetic. The Evans method uses this relationship to obtain the magnetic susceptibility, and thus the magnetic moment, of that paramagnetic solute.
An Evans method sample uses a capillary insert containing a mixture of a deuterated solvent and the matching proteated solvent. The compound of interest is dissolved in the same solvent mixture and placed in an NMR tube with the capillary.
The acquired NMR spectrum shows two solvent peaks: one corresponding to the proteated solvent in solution with the compound, and the other corresponding to the proteated solvent in the capillary.
The magnetic susceptibility is calculated from the frequency difference and the concentration of the paramagnetic compound in the sample.
The magnetic moment is calculated from the magnetic susceptibility in a special unit called the Bohr magneton. The magnetic moment can then be compared to theoretical spin-only values to estimate the number of unpaired electrons in the sample.
Now that you understand the principles of the Evans method, let’s go through a procedure for finding the number of unpaired electrons in Fe(acac)3 with the Evans method.
To prepare the capillary insert, melt the tip of a long Pasteur pipette with a flame until the tip melts into a glass bulb. Allow the glass to cool.
Next, combine in a clean scintillation vial 2 mL of a deuterated solvent and 40 μL of a proteated solvent. Cap the vial and swirl gently.
Carefully add a few drops of the solvent mixture to the cooled pipette. Gently flick or tap the pipette tip until the solvent has gathered at the bottom of the tip.
Continue adding the solvent mixture in this way until the solution fills the sealed pipette tip to a depth of about 2 inches, with no air bubbles.
Cap the pipette with a 14/20 rubber septum. Equip a 3-mL syringe with a needle. Insert the needle through the septum and carefully withdraw 3 mL of air.
Remove the syringe and clamp the pipette to a ring stand horizontally. Use a lighter to soften the glass above the solution in the pipette tip.
Once the glass begins softening, slowly rotate the solution-filled pipette tip to seal in the solution. Continue rotating the newly-formed capillary until it easily separates from the pipette body.
Let the capillary insert cool, and then store it in a labeled container.
To prepare a sample for the Evans method, first record the mass of a scintillation vial and cap. Then, place 5 mg of the paramagnetic compound of interest into the scintillation vial and record the mass.
Pipette about 600 μL of the mixture of deuterated and proteated solvents into the scintillation vial. Swirl the vial until the solid compound completely dissolves.
Record the mass of the capped vial of sample solution. Then, obtain a standard NMR tube and cap.
Carefully slide the capillary insert into the NMR tube at an angle. Transfer the solution of the paramagnetic compound to the NMR tube and cap the tube. Ensure that the insert is sitting at the bottom of the tube.
Acquire and save a standard 1H NMR spectrum.
First, calculate the concentration of the sample solution in moles per cubic centimeter using the recorded masses and the density of the solvent. Then, convert the difference between the solvent peak chemical shifts from ppm to Hz. Calculate the molar magnetic susceptibility of the sample.
Next, calculate the magnetic moment from the probe temperature and the molar magnetic susceptibility. Compare the calculated value with a table of known values to determine the number of unpaired electrons in the compound.
The number of unpaired electrons is important for modeling chemical and biological complexes. Let’s look at a few applications.
Transition metal complexes can be modeled with molecular orbital theory. In this model, electrons are assigned to molecular orbitals shared between atoms. Information about the number of unpaired electrons helps to confirm that an appropriate model is being used. Further, the number of singly-occupied and unoccupied orbitals predicts how the complex will react with other molecules.
Molecules can be classified by the symmetry operations that they can perform, such as being mirrored across an axis. Molecular symmetry can predict many properties, such as the vibrational modes of a compound. As the number of unpaired electrons can provide information about molecular geometry, it is important to accurately determine the number of unpaired electrons when characterizing compounds.
You’ve just watched JoVE’s introduction to the Evans method. You should now understand the underlying principles of the Evans method, the procedure for calculating the number of unpaired electrons, and how unpaired electrons are relevant to understanding chemical reactivity. Thanks for watching!
