Die Evans-Methode
English
COMPARTILHAR
Visão Geral
Quelle: Tamara M. Powers, Department of Chemistry, Texas A & M Universität
Während die meisten organischen Moleküle diamagnetisch sind, Staaten wobei alles, was ihre Elektronen in Anleihen, viele Übergang Metall komplexe gepaart sind paramagnetisch, sind die Erden hat mit ungepaarten Elektronen. Rückrufaktion Hund’sche Regel, die besagt, dass für orbitale ähnliche Energien, Elektronen die orbitale maximiert die Zahl der ungepaarten Elektronen vor pairing füllen werden. Übergangsmetalle haben teilweise aufgefüllt d-orbitale, dessen Energien sind in unterschiedlichem Ausmaß durch Koordinierung der Liganden an das Metall gestört. So, die d-orbitale sind in Energie einander ähnlich, jedoch nicht alle entartet. Dies ermöglicht eine komplexe diamagnetisch, mit alle Elektronen gepaart oder paramagnetisch mit ungepaarten Elektronen.
Zu wissen, die Zahl der ungepaarten Elektronen in einem Metall komplexe kann Hinweise geben in der Oxidationsstufe und der Geometrie des Metalls komplexe sowie in die Liganden (Kristallfeld) Feldstärke der Liganden. Diese Eigenschaften einen enormen Einfluss auf die Spektroskopie und Reaktivität der Übergang Metall komplexe und sind also wichtig, zu verstehen.
Eine Möglichkeit, die Anzahl der ungepaarten Elektronen wird zur Messung der magnetischen Suszeptibilität χ, der Koordinierung Verbindung. Magnetische Suszeptibilität ist das Maß der Magnetisierung der ein Material (oder Verbindung), wenn in einem angelegten Magnetfeldes platziert. Gepaarte Elektronen sind leicht durch einen angelegten Magnetfeldes abgestoßen, und diese Abstoßung steigt linear als die Stärke des Magnetfeldes erhöht. Auf der anderen Seite ungepaarte Elektronen sind ein Magnetfeld (in größerem Umfang) angezogen, und die Attraktivität steigt linear mit der magnetischen Feldstärke. Daher wird eine Verbindung mit ungepaarten Elektronen ein Magnetfeld angezogen werden. 1
Wenn wir der magnetischen Suszeptibilität messen, erhalten wir Informationen über die Zahl der ungepaarten Elektronen aus das magnetische Moment μ. Die magnetische Suszeptibilität bezieht sich auf das magnetische Moment μ von Gleichung 12:
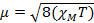 (1)
(1)
Die Konstante 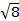 = [(3kB) / Nβ2)], wo β = Bohr Magneton des Elektrons (0,93 X 10-20 Erg Gauß-1), N = Avogadro’s Zahl und kB = Boltzmann-Konstante
= [(3kB) / Nβ2)], wo β = Bohr Magneton des Elektrons (0,93 X 10-20 Erg Gauß-1), N = Avogadro’s Zahl und kB = Boltzmann-Konstante
XM = molare magnetische Suszeptibilität (cm3/mol)
T = Temperatur (K)
µ = magnetisches Moment, gemessen in Einheiten von Bohr Magneton, µB = 9,27 x 10-24 JT-1
Das magnetische Moment für komplexe ist gegeben durch die Gleichung 21:
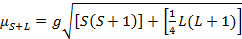 (2)
(2)
g = Gyromagnetic Verhältnis = 2.00023 µB
S = Spin-Quantenzahl = ∑ms = [Anzahl der ungepaarten Elektronen, n] / 2
L = orbital Quantenzahl = ∑ml
Diese Gleichung hat Orbital und Spin Beiträge. Für die erste Zeile Übergang Metall komplexe, die orbitale Beitrag ist klein und daher kann weggelassen werden, damit das Drehen nur magnetische Moment gegeben ist Gleichung 3:
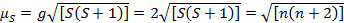 (3)
(3)
Das Spin-nur magnetische Moment kann somit direkt die Zahl der ungepaarten Elektronen geben. Diese Annäherung kann auch für schwerere Metalle erfolgen, obwohl orbital Beiträge für zweite und dritte Reihe Übergangsmetallen wichtig sein könnten. Dieser Beitrag kann so bedeutend, dass es das magnetische Moment genug, das die Verbindung mehr Elektronen ungepaarten haben bläst scheint, als dies der Fall ist. Daher möglicherweise zusätzliche Charakterisierung für diese komplexe erforderlich.
In diesem Experiment wird die Lösung magnetisches Moment des tris(acetylacetonato)iron(III) (Fe(acac)3) bestimmt experimentell mit Evans Methode in Chloroform.
Princípios
Procedimento
Resultados
Experimental Results
| Fe(acac)3 | Chloroform | |
| m (g) | 0.0051 | 0.874 |
| MW (g/mol) | 353.17 | n/a |
| n (mol) | 1.44⋅10–5 | n/a |
| Density (g/mL) | n/a | 1.49* |
| Volume (mL) | n/a | 0.587 |
| c (mol/mL) | 2.45⋅10-5 | |
| NMR shifts | Peak 1 | Peak 2 |
| δ (ppm) | 7.26 | 5.85 |
| Δppm | 1.41 | |
| NMR Instrument | ||
| Temperature (K) | 296.3 | |
| Field, F (Hz) | 500⋅106 |
* the density of the solvent can be approximated to the density of the solvent used
Calculations:

 = 0.0137 cm3/mol
= 0.0137 cm3/mol
 = 5.70 µB
= 5.70 µB
Theoretical Results for Given S and n Values:
| S | n | μS |
| 1/2 | 1 | 1.73 |
| 1 | 2 | 2.83 |
| 3/2 | 3 | 3.87 |
| 2 | 4 | 4.90 |
| 5/2 | 5 | 5.92 |
For 4.5 mg of Fe(acac)3 dissolved in 0.58 mL solvent, with a 300 MHz instrument a peak separation of 1.41 ppm is observed, which gives XM= 1.37 x 10-2 and µeff = 5.70. This µeff value is consistent with an S = 5/2 complex, which has 5 unpaired electrons.
Applications and Summary
The Evans method is a simple and practical method for obtaining the magnetic susceptibility of soluble metal complexes. This provides the number of unpaired electrons in a metal complex, which is pertinent to the spectroscopy, magnetic properties, and reactivity of the complex.
Measuring the magnetic susceptibility of paramagnetic species gives the number of unpaired electrons, which is a key property of metal complexes. As the reactivity of metal complexes is influenced by its electronic structure – that is, how the d-orbitals are populated – it is important to establish the number of unpaired electrons. The magnetic susceptibility can be used to determine the geometry of the metal complex in solution, give insight into the ligand field strength, and can provide evidence for the correct formal oxidation-state assignment of the metal complex. In the modules on "Group Theory" and "MO Theory of Transition Metal Complexes," we will introduce how to predict d-orbital splitting diagrams as well as how to use data from the Evans method to help determine the geometry of a metal complex and provide evidence for the oxidation state of the metal center.
There are multiple instruments that can be used to measure the magnetic susceptibility of a paramagnetic species including a Gouy balance, SQUID, or NMR instrument. The Evans method is a simple and practical technique that uses NMR to determine the solution magnetic moment of a paramagnet. While the Evans method is a powerful tool in the field of magnetism, there are several drawbacks to the technique. First, the molecule must be soluble in the solvent used in the experiment. If the paramagnetic sample is not fully dissolved, the concentration of the solution will be incorrect, which will lead to errors in the experimentally determined solution magnetic moment. Other errors in concentration can arise if the paramagnetic sample has diamagnetic (solvent) or paramagnetic impurities.
Referências
- Miessler, G. L., Fischer, P. J., Tarr, D. A. Inorganic Chemistry. 5 ed. Pearson. (2014).
- Drago, R. S. Physical Methods for Chemists. 2 ed. Saunders College Publishing. (1992).
- Girolami, G. S., Rauchfuss, T. B., Angelici, R. J. Synthesis and Technique in Inorganic Chemistry: A Laboratory Manual. 3 ed. University Science Books. Sausalito, CA, (1999).
Transcrição
The Evans method is a technique for calculating the number of unpaired electrons in solution-state metal complexes.
Many transition metal complexes have unpaired electrons, making them attracted to magnetic fields. These complexes are called paramagnetic. Complexes with all paired electrons are called diamagnetic.
Knowing the number of unpaired electrons is important for predicting the reactivity of a compound. The Evans method uses NMR spectroscopy to measure the parameters needed to calculate the number of unpaired electrons.
This video will illustrate the procedure for performing the Evans method, demonstrate the analysis of Fe(acac)3, and introduce a few applications of counting unpaired electrons in chemistry.
The number of unpaired electrons in a complex can be determined from the magnetic moment of the given molecule. The magnetic moments of 1st row transition metal complexes can be approximated from the contributions of unpaired electrons, called the spin-only magnetic moment. For the 2nd and 3rd row transition metal complexes, both the spin and orbital contributions must be considered.
The magnetic moment is related to the magnetic susceptibility, which provides the degree of magnetization of a complex in an applied magnetic field.
The chemical shift of a species in an NMR spectrum is affected by the overall magnetic susceptibility of the sample solution. Thus, the chemical shift of a solvent changes if the solute is paramagnetic. The Evans method uses this relationship to obtain the magnetic susceptibility, and thus the magnetic moment, of that paramagnetic solute.
An Evans method sample uses a capillary insert containing a mixture of a deuterated solvent and the matching proteated solvent. The compound of interest is dissolved in the same solvent mixture and placed in an NMR tube with the capillary.
The acquired NMR spectrum shows two solvent peaks: one corresponding to the proteated solvent in solution with the compound, and the other corresponding to the proteated solvent in the capillary.
The magnetic susceptibility is calculated from the frequency difference and the concentration of the paramagnetic compound in the sample.
The magnetic moment is calculated from the magnetic susceptibility in a special unit called the Bohr magneton. The magnetic moment can then be compared to theoretical spin-only values to estimate the number of unpaired electrons in the sample.
Now that you understand the principles of the Evans method, let’s go through a procedure for finding the number of unpaired electrons in Fe(acac)3 with the Evans method.
To prepare the capillary insert, melt the tip of a long Pasteur pipette with a flame until the tip melts into a glass bulb. Allow the glass to cool.
Next, combine in a clean scintillation vial 2 mL of a deuterated solvent and 40 μL of a proteated solvent. Cap the vial and swirl gently.
Carefully add a few drops of the solvent mixture to the cooled pipette. Gently flick or tap the pipette tip until the solvent has gathered at the bottom of the tip.
Continue adding the solvent mixture in this way until the solution fills the sealed pipette tip to a depth of about 2 inches, with no air bubbles.
Cap the pipette with a 14/20 rubber septum. Equip a 3-mL syringe with a needle. Insert the needle through the septum and carefully withdraw 3 mL of air.
Remove the syringe and clamp the pipette to a ring stand horizontally. Use a lighter to soften the glass above the solution in the pipette tip.
Once the glass begins softening, slowly rotate the solution-filled pipette tip to seal in the solution. Continue rotating the newly-formed capillary until it easily separates from the pipette body.
Let the capillary insert cool, and then store it in a labeled container.
To prepare a sample for the Evans method, first record the mass of a scintillation vial and cap. Then, place 5 mg of the paramagnetic compound of interest into the scintillation vial and record the mass.
Pipette about 600 μL of the mixture of deuterated and proteated solvents into the scintillation vial. Swirl the vial until the solid compound completely dissolves.
Record the mass of the capped vial of sample solution. Then, obtain a standard NMR tube and cap.
Carefully slide the capillary insert into the NMR tube at an angle. Transfer the solution of the paramagnetic compound to the NMR tube and cap the tube. Ensure that the insert is sitting at the bottom of the tube.
Acquire and save a standard 1H NMR spectrum.
First, calculate the concentration of the sample solution in moles per cubic centimeter using the recorded masses and the density of the solvent. Then, convert the difference between the solvent peak chemical shifts from ppm to Hz. Calculate the molar magnetic susceptibility of the sample.
Next, calculate the magnetic moment from the probe temperature and the molar magnetic susceptibility. Compare the calculated value with a table of known values to determine the number of unpaired electrons in the compound.
The number of unpaired electrons is important for modeling chemical and biological complexes. Let’s look at a few applications.
Transition metal complexes can be modeled with molecular orbital theory. In this model, electrons are assigned to molecular orbitals shared between atoms. Information about the number of unpaired electrons helps to confirm that an appropriate model is being used. Further, the number of singly-occupied and unoccupied orbitals predicts how the complex will react with other molecules.
Molecules can be classified by the symmetry operations that they can perform, such as being mirrored across an axis. Molecular symmetry can predict many properties, such as the vibrational modes of a compound. As the number of unpaired electrons can provide information about molecular geometry, it is important to accurately determine the number of unpaired electrons when characterizing compounds.
You’ve just watched JoVE’s introduction to the Evans method. You should now understand the underlying principles of the Evans method, the procedure for calculating the number of unpaired electrons, and how unpaired electrons are relevant to understanding chemical reactivity. Thanks for watching!
